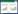2. 广西医科大学附属武鸣医院科研部, 南宁 530199
2. Research Department, Affiliated Wuming Hospital of Guangxi Medical University, Nanning 530199, China
脓毒症(Sepsis)是对感染应答失调而导致的危及生命的急性器官功能障碍综合征[1]。25%~50%的脓毒症患者会出现急性肺损伤(acute lung injury, ALI),病死率高达40%[2]。临床表现为顽固性低氧血症和呼吸窘迫为特征的全身炎症反应[3]。目前脓毒症诱导的ALI治疗重点仍以控制感染、恢复灌注和呼吸支持为主,缺乏相关特效药物[4]。当前普遍认为脓毒症ALI的病理生理机制与促炎和抗炎系统之间的不平衡引起的炎症反应有关,持续性的过度炎症反应可损伤肺泡毛细血管和上皮细胞[5]。持续性的过度炎症反应在脓毒症ALI中发挥着重要作用,但具体的产生机制目前仍不明确。
泛凋亡是一种独特的、生理学相关的、炎性程序性细胞死亡通路,同时具有细胞焦亡、细胞凋亡和坏死性凋亡的特征[6]。在感应到病原体成分后,其传感器蛋白就会介导其他蛋白质组装成泛凋亡小体复合物,从而诱导泛凋亡,引发机体炎性细胞死亡[7]。泛凋亡与持续性炎症和病理损伤有关[8]。目前有研究证实泛凋亡在ALI中的重要作用[9]。AIM2炎症小体的激活可以促进泛凋亡,进而加剧持续性炎症反应的发生[10]。近些年研究发现,JAK/STAT1通路是驱动细胞焦亡、凋亡和坏死性凋亡(泛凋亡)的发生所必须的[11]。然而,AIM2炎症小体是否通过JAK2/STAT1信号通路促进ZBP1-PANoptosis途径加剧脓毒症ALI尚未见报道。
本研究通过抑制AIM2炎症小体观察其对小鼠肺部病理和相关标志物表达的影响;通过抑制JAK2/STAT1通路表达观察细胞死亡、线粒体损伤和相关标志物表达等情况,旨在阐明AIM2炎症小体通过激活JAK2/STAT1信号通路促进ZBP1-PANoptosis途径加重脓毒症ALI的作用机制。
1 材料与方法 1.1 脓毒症ALI模型和处理雄性C57BL/6小鼠,7~8周,购自斯贝福(北京)生物技术有限公司。将小鼠安置在广西医科大学动物中心SPF级实验室,温度控制(24±2)℃无特定病原体的设施中,光照/黑暗循环为12 h,可自由获取食物和水。动物实验由广西医科大学实验动物福利与伦理委员会审查批准(批号:202502011)。在实验之前,使小鼠适应环境1周。所有小鼠按照随机数字法随机分为3组(每组6只):正常组(NC)、LPS模型组、AIM2炎症小体抑制剂(A151+LPS)组。造模方式为通过腹腔注射,向小鼠注射LPS(10 mg/kg)(HY-D1056,MCE,US)。A151+LPS组使用相同的方式造模,并随后立即注射A151 (0.015 g/kg,i.p) (HY-150751C,MCE,US)。在造模完成24 h后,使用戊巴比妥钠(100 mg/kg,i.p)对小鼠实施安乐死,取肺组织备检。
1.2 肺泡灌洗液(bronchoalveolar lavage fluid,BALF)的收集和分析使用注射器收集BALF,将1 mL冰冷的磷酸盐缓冲液(PBS)气管内滴注到肺中,然后灌洗3次。将支气管BALF以1 500 r/min离心10 min,并将BALF上清液储存在-80℃用于进一步检测。使用BCA蛋白测定试剂盒(HBCA,HYCEZMBIO,China)测定BALF蛋白浓度。根据制造商的说明,使用小鼠TNF-α酶联免疫测定试剂盒(88-7324,Invitrogen,US)、小鼠IL-1β酶联免疫测定试剂盒(88-7013,Invitrogen,US)和小鼠IL-6酶联免疫测定试剂盒(88-7064, ,Invitrogen,US)检测TNF-α、IL-1β和IL-6的水平。
1.3 Hematoxylin-eosin(HE)染色将肺组织固定在4%聚甲醛(Servicebio,G1101)中,然后脱水并包埋在石蜡中。切割组织切片并进行HE染色。
1.4 Western Blot使用与蛋白酶抑制剂(Thermo)、磷酸化蛋白酶抑制剂(Servicebio)预混合的总蛋白提取缓冲液提取总蛋白,并使用BCA蛋白测定试剂盒定量。通过10%或12%十二烷基硫酸钠-聚丙烯酰胺凝胶电泳分离等量的蛋白质,并转移到聚偏二氟乙烯膜上。在室温下非磷酸化蛋白用5%新鲜脱脂牛奶(用TBST配制)中封闭1 h,磷酸化蛋白在无蛋白封闭液(Shrebio)中封闭30 min。然后与抗体IGF2BP1(R24661, ZENBIO, China),Total and cleaved N-terminal GSDMD Antibody(P30823, ABMART, China),GSDME + N-terminal Antibody(P79886, ABMART, China),Caspase 3/P17/P19(19677-1-AP, Proteintech, China),Caspase-8/P43/P18(13423-1-AP, Proteintech, China),cleaved caspase-8(8592T, CST, China),MLKL(R380559, ZENBIO, China),Phospho-MLKL L(R382136, ZENBIO, China),Phospho-MLKL L(R30265, ZENBIO, China),RIPK3(PA4203, ABMART, China),P-RIPK3(PJP12295, ABMART, China),, JAK2(R24775, ZENBIO, China),Phospho-JAK2(ab32101,Abcam,China),STAT1(R25799, ZENBIO, China),Phospho-STAT1(ab278713, Abcam, China)上述抗体稀释比均为1∶1000,β-ACTIN(66009-1-Ig, Proteintech, 1∶5000)在4℃下孵育过夜。用TBST洗涤3次后,将膜与Goat Anti-Rabbit IgG H & L(HRP)(511203, ZENBIO, 1∶5 000)在室温下孵育1 h。然后使用扫描成像仪使膜可视化。
1.5 细胞培养和处理THP-1购自海星生物,并培养在37℃,5% CO2的加湿培养箱中和维持在含有10%胎牛血清和1%青霉素-链霉素溶液的1640培养基中。实验分为3组:NC组、LPS(1 μg/mL)组、Ruxolitinib(5 μmol/L)+LPS(1 μg/mL)。将细胞接种在六孔板(1×106/孔),使用佛波酯(PMA,100 ng/mL)24 h将THP-1诱导为M0巨噬细胞,Ruxolitinib+LPS(1 μg/mL)组处理方式是在加入LPS前2 h加入Ruxolitinib(5 μmol/L)进行预处理。24 h后提取RNA,48 h后提取蛋白。
1.6 Calcein-AM/PI活细胞/死细胞双染色将低温保存Calcein-AM/PI试剂盒(G1707-100T,Servicebio,China)取出并恢复到室温,按说明书配置工作液。对于贴壁细胞,去除上清液,使用PBS充分洗涤细胞3次。加入上述Calcein AM/PI检测工作液,于细胞培养箱中,避光孵育15 min。使用荧光显微镜对活细胞和死细胞进行观察分析。
1.7 透射电镜弃培养基,加入电镜固定液,常温固定5 min;用细胞刮将细胞轻柔刮下,离心收集细胞,弃固定液,加入新的固定液重悬,室温避光固定30 min;4℃保存。离心收集细胞,弃上清液,加入0.1 mol/L磷酸缓冲液混匀漂洗3 min后再离心,重复3次;用1%琼脂糖溶液预包埋,1%锇酸避光室温固定2 h,漂洗3次;室温进行酒精脱水处理;渗透包埋;包埋板放于60℃烤箱聚合48 h,取出树脂块备用;树脂块于超薄切片机60~80 nm超薄切片,150目方华膜铜网捞片;铜网进行染色处理;透射电子显微镜下观察,采集图像分析。
1.8 Real-time reverse transcriptase-PCR(RT-qPCR)使用Trizol试剂提取总RNA,并使用ToloScript All-in-one RT EasyMix for Qpcr(22107,TOLOBIO,China)逆转录成cDNA。使用Q1 SYBR qPCR Master Mix (Universal)(22201,TOLOBIO,China)根据制造商的说明在QuantStudio 3实时PCR系统上进行cDNA扩增,所有引物均来自中国生工。采用比较Ct法(2-ΔΔCT)分析数据。
1.9 统计学方法使用Graph Pad Prism 10.1.2软件进行统计分析。数据表示为均数±标准差(x±s)。计量资料符合正态分布同时方差齐时,用单因素方差分析进行多组间比较,用Dunnett多重比较检验三组之间的差异。不符合正态分布或方差不齐时,用非参数检验进行组间比较。以P < 0.05为差异有统计学意义。
2 结果 2.1 各组小鼠肺组织病理检查结果和肺泡灌洗液炎症因子的变化HE结果显示,NC组肺组织无明显形态学损伤,无明显炎症细胞浸润和肺泡间隔增厚。LPS组肺组织存在明显的形态学损伤,包括炎性细胞浸润和肺泡间隔增厚,而这些损伤在A151+LPS组明显减轻,表现为炎症细胞浸润和肺泡间隔增厚情况减轻。ELISA结果显示,与NC组相比,LPS组TNF-α、IL-1β、IL-6的表达水平显著升高, 差异有统计学意义(均P < 0.05)。与LPS组相比,A151+LPS组TNF-α、IL-1β、IL-6的表达水平降低(P < 0.05,见图 1)。
|
| 注:A为各组小鼠肺组织HE染色(×40);B为各组小鼠BALF中TNF-α的含量;C为各组小鼠BALF中IL-1β的含量;D为各组小鼠BALF中IL-6的含量;vs. LPS,aP < 0.05, bP < 0.01, cP < 0.01, dP < 0.01 图 1 各组小鼠肺组织病理检查结果和肺泡灌洗液炎症因子情况(HE×40,n=6) Fig 1 Pathological examination results of lung tissue and inflammatory factors of alveolar lavage fluid of mice in each group (HE×40, n=6) |
|
|
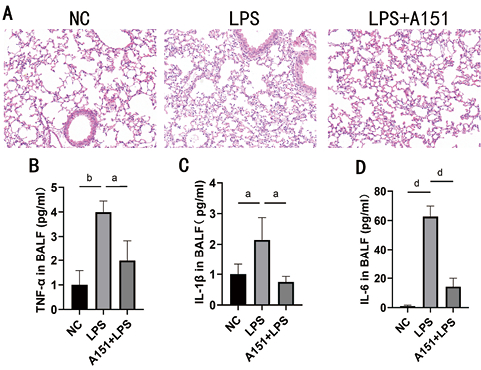
Western Blot检测结果显示,与NC组相比,LPS组小鼠肺组织ZBP1、GSDMD-N、GSDME-N、caspase3 P17、caspase-8 P18、t/p-MLKL表达显著增多意义(均P < 0.05),而t/p-RIPK3蛋白表达差异无统计学意义(P > 0.05)。与LPS组相比,A151+LPS组ZBP1、GSDMD-N、GSDME-N、caspase3 P17、caspase-8 P18、t/p-MLKL表达显著减少(均P < 0.05),但是t/p-RIPK3蛋白表达差异无统计学意义(P > 0.05,见图 2)。
|
| 注:A为各组小鼠肺组织中ZBP1、GSDMD-F+N、GSDME-F+N蛋白表达情况;B为各组小鼠肺组织中Caspase8 P43/P18、Caspase3 P32/P17蛋白表达情况;C为各组小鼠肺组织中t/p-RIPK3、t/p-MLKL蛋白表达情况;D-F分别为A、B、C的量化图;vs. LPS,aP < 0.05, bP < 0.01, cP < 0.001, dP < 0.0001 图 2 Western Blot检测各组小鼠肺组织ZBP1-PANoptosis蛋白标志物表达情况(n=6) Fig 2 Western blot was used to detect the expression of ZBP1-PANoptosis protein markers in lung tissue of mice in each group(n=6) |
|
|
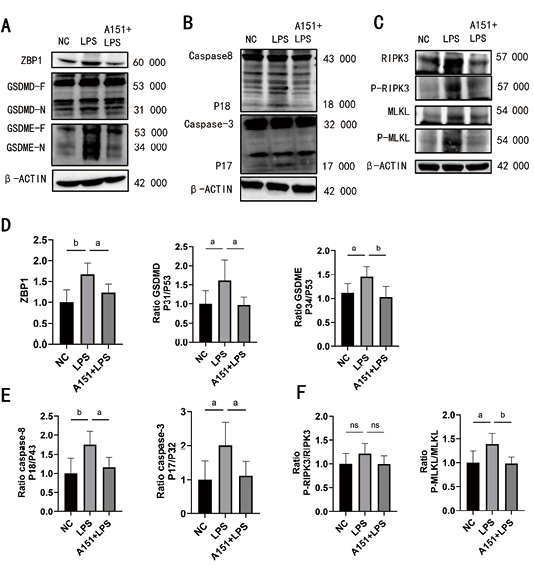
Western Blot检测结果显示,与NC组相比,LPS组p-JAK2、p-STAT1蛋白表达升高(P < 0.01)。与LPS组相比,A151+LPS组p-JAK2、p-STAT1蛋白表达下降(P < 0.05)。见图 3。
|
| 注:图A为各组小鼠肺组织中JAK2、p-JAK2、STAT1、p-STAT1蛋白表达情况;图B为各组小鼠肺组织中JAK2、p-JAK2、STAT1、p-STAT1蛋白表达量化;vs. LPS,aP < 0.05, bP < 0.01, cP < 0.01, dP < 0.01 图 3 Western Blot检测各组小鼠肺组织JAK2/STAT1信号通路变化(n=6) Fig 3 Western blot was used to detect the changes of JAK2/STAT1 signaling pathway in lung tissue of mice in each group(n=6) |
|
|
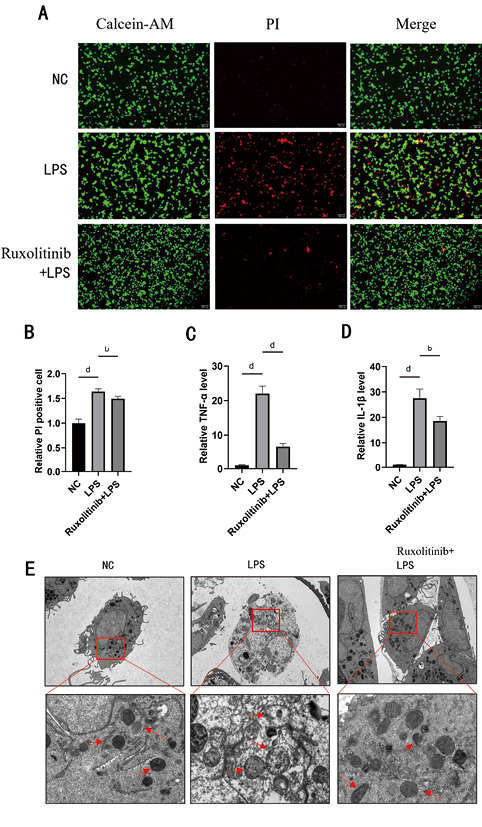
Calcein-AM/PI细胞染色结果显示,与NC组相比,LPS组细胞病死率升高(P < 0.01)。与LPS组相比,Ruxolitinib+LPS组细胞病死率降低(P < 0.01)。RT-qPCR结果显示,与NC组相比,LPS组TNF-α和IL-1β的mRNA表达明显升高(P < 0.01)。而Ruxolitinib+LPS组TNF-α和IL-1β的mRNA表达水平低于LPS组(P < 0.01)。透射电镜结果显示LPS组细胞膜完整性被破坏,细胞质内容物外渗,线粒体肿胀,基质呈现空泡化,嵴断裂消失;Ruxolitinib+LPS组细胞膜完整性较好,线粒体结构虽然完整,但嵴变短,数目减少,排列紊乱,肿胀(见图 4)。
|
| 注:A为各组细胞的Calcein-AM/PI细胞染色的免疫荧光结果图,Calcein-AM为活细胞染色(绿色荧光),PI为死细胞染色(红色荧光);B为各组细胞Calcein-AM/PI细胞染色的免疫荧光结果量化;C为各组细胞TNF-αmRNA表达水平;D为各组细胞IL-1βmRNA表达水平;E为各组细胞透射电镜结果;vs. LPS,aP < 0.05, bP < 0.01, cP < 0.01, dP < 0.01 图 4 Calcein-AM/PI、RT-qPCR和透射电镜检测细胞损伤和死亡情况(Calcein-AM/PI 50μmol/L,n=6,TEM ×3.0K,×15.0K) Fig 4 Calcein-AM/PI, RT-qPCR, and transmission electron microscopy detect cell damage and death(Calcein-AM/PI 50μmol/L, n=6, TEM ×3.0K, ×15.0K) |
|
|
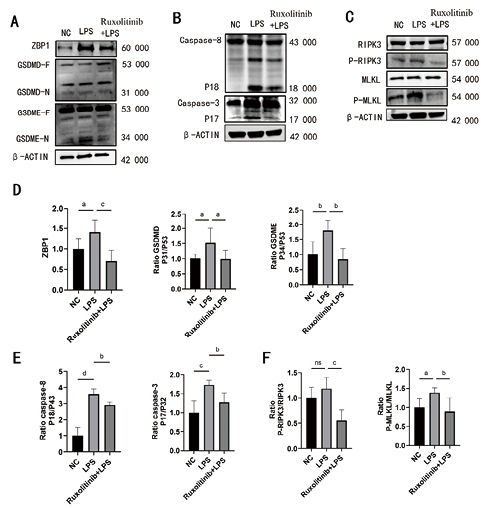
Western Blot结果显示,与NC组相比,LPS组细胞中ZBP1、GSDMD-N、GSDME-N、caspase3 P17、caspase-8 P18、p-RIPK3、p-MLKL的表达增多(均P < 0.05),但t/p-RIPK3差异无统计学意义(P > 0.05)。与LPS组相比,Ruxolitinib+LPS组中ZBP1、GSDMD-N、GSDME-N、caspase3 P17、caspase-8 P18、p-RIPK3、p-MLKL的表达减少(均P < 0.05,见图 5)。
|
| 注:A为各组细胞中ZBP1、GSDMD-F+N、GSDME-F+N蛋白表达情况;B为各组细胞中Caspase8 P43/P18、Caspase3 P32/P17蛋白表达情况;C为各组细胞中t/p-RIPK3、t/p-MLKL蛋白表达情况;D-F分别为A、B、C的量化图;vs. LPS,aP < 0.05, bP < 0.01, cP < 0.01, dP < 0.01 图 5 Western Blot检测各组细胞ZBP1-PANoptosis蛋白标志物表达情况(n=6) Fig 5 Western blot was used to detect the expression of ZBP1-PANoptosis protein markers in each group(n=6) |
|
|
脓毒症所致ALI的并发症发生率和病死率高于其他病因所致的ALI患者, 肺内失控的过度炎症反应被认为是脓毒症ALI的重要特征[12]。因此,进一步明确脓毒症所致肺损伤中肺组织发生过度炎症反应的分子机制对于靶向药物的研发是至关重要的。
AIM2炎症小体由AIM2、凋亡相关斑点样蛋白(含CARD结构域)(Apoptosis-associated speck-like protein containing a CARD,ASC)、半胱天冬蛋白酶1前体(pro-caspase-1)组成[13]。AIM2可识别多种细菌和病毒病原体的双链DNA(dsDNA),在各种疾病中都发挥着重要作用,包括传染病、炎症性疾病和癌症[14]。在脓毒症中,AIM2对脓毒症诊断具有潜在诊断价值[15]。研究报道AIM2可结合ASC、caspase-8和caspase-1形成炎性复合物,进而触发细胞凋亡和细胞焦亡[16]。细胞焦亡和凋亡均可以加剧ALI中的炎性细胞死亡反应[17-18]。近几年有研究证实AIM2介导的信号转导可作为含NLR家族Pyrin域蛋白1(PYRIN)和ZBP1的上游调节因子以诱导和调控泛凋亡(细胞焦亡、凋亡、坏死性凋亡),介导炎症细胞死亡和细胞因子释放[19]。相关炎症因子和信号通路的激活可促使机体产生一系列免疫应答,诱导肺部炎性损伤[20]。研究报道抑制AIM2炎症小体可降低泛凋亡的发生,进而减轻脓毒症诱导的肾损伤[21]。本研究发现抑制AIM2炎症小体活性后脓毒症诱导的肺组织损伤被改善和炎症介质分泌减少,ZBP1-PANoptosis标志物的表达下降,但p-RIPK3/t-RIPK3无组间差异,分析原因可能为不同小鼠的代谢状态和肺组织免疫细胞浸润存在差异,进而影响RIPK3表达,对结果造成了干扰。这些结果提示AIM2炎症小体可能通过调节ZBP1-PANoptosis以促进脓毒症ALI发生。那么AIM2炎症小体又是如何驱动ZBP1-PANoptosis发生的呢?
研究报道JAK2/STAT1通路在脓毒症ALI的肺组织炎症细胞死亡中发挥重要作用[22]。临床上已有一些STAT抑制剂和JAK抑制剂被用于炎性疾病的治疗[23-24]。本研究发现LPS诱导的脓毒症ALI小鼠肺组织中p-JAK2、p-STAT1表达上调。干扰素调节因子(IRF1)一方面受到JAK/STAT1通路调节,参与炎性细胞死亡,另一方面通过促进STAT1的DNA结合,参与JAK/STAT信号的正反馈环,引发过度的炎症反应[25-26]。研究发现敲除AIM2可抑制IRF1的表达,减轻炎症反应。根据以上研究结果推测AIM2炎症小体可能通过IRF1调控JAK2/STAT1通路,进而影响肺部炎症损伤[27]。虽然尚无报道指出AIM2炎症小体是否参与调控JAK2/STAT1通路的表达,但依据笔者团队前期的实验结果,做了进一步的探讨。本研究发现抑制AIM2炎症小体可下调p-JAK2、p-STAT1的表达,而JAK2、STAT1水平没有变化,提示AIM2炎症小体可能参与调控JAK2/STAT1通路的磷酸化过程。
JAK2/STAT1信号转导是导致多种炎症性疾病的常见通路,主要通过炎症反应失控、肺泡-毛细血管屏障破坏及氧化应激加剧等机制诱导ALI[28]。临床研究发现该通路激活可促使重症病毒性肺炎患者发生不可逆的炎性损伤[26]。而抑制JAK2/STAT1通路可降低TNF-α和IL-1β、IL-18表达,改善LPS诱导的肺组织损伤[29]。JAK/STAT1/IRF1通路在泛凋亡的发生发展中发挥重要作用[30]。本研究发现LPS可诱导ZBP1-PANoptosis表达上调,抑制JAK2/STAT1通路可下调ZBP1-PANoptosis标志物表达,细胞病死率和TNF-α、IL-1β转录水平也明显下降,且透射电镜结果显示细胞损伤状态被改善。这些结果显示JAK2/STAT1通路可能是驱动ZBP1-PANoptosis的关键通路之一。
综上所述,本研究证明了AIM2炎症小体通过JAK2/STAT1通路促进ZBP1-PANoptosis在脓毒症ALI中的新机制,为脓毒症ALI更深层次的机制探索和靶向药物研发提供了新方向。本研究仍具有一定的局限性,脓毒症ALI涉及多种炎症通路和因子分泌,本研究仅对其中一条机制进行探究,因此针对其他可能机制及其之间的关联的研究是必要的;在客观因素的限制下,本研究缺乏了过表达或激动剂作为对照,对于AIM2炎症小体是否直接调控JAK2/STAT1通路仍有待进一步探究。
利益冲突 所有作者声明无利益冲突
作者贡献声明 陈雪怡:实验设计、实验操作、数据分析,论文撰写;张凌峰:实验操作,数据分析;张景鸿:实验设计,论文修改、支持性贡献
| [1] | Meyer NJ, Prescott HC. Sepsis and septic shock[J]. N Engl J Med, 2024, 391(22): 2133-2146. DOI:10.1056/NEJMra2403213 |
| [2] | 鲁俊才, 李政玉, 张文星, 等. 脓毒症相关急性肺损伤发病机制及新型生物标志物的研究进展[J]. 中国临床医生杂志, 2025, 53(6): 695-699. DOI:10.3969/j.issn.2095-8552.2025.06.008 |
| [3] | Auriemma CL, Zhuo HJ, Delucchi K, et al. Acute respiratory distress syndrome-attributable mortality in critically ill patients with sepsis[J]. Intensive Care Med, 2020, 46(6): 1222-1231. DOI:10.1007/s00134-020-06010-9 |
| [4] | Jin C, Chen J, Gu J, et al. Gut-lymph-lung pathway mediates sepsis-induced acute lung injury[J]. Chin Med J, 2020, 133(18): 2212-2218. DOI:10.1097/CM9.0000000000000928 |
| [5] | Lin SH, Zhao YS, Zhou DX, et al. Coronavirus disease 2019 (COVID-19): cytokine storms, hyper-inflammatory phenotypes, and acute respiratory distress syndrome[J]. Genes Dis, 2020, 7(4): 520-527. DOI:10.1016/j.gendis.2020.06.009 |
| [6] | Samir P, Subbarao Malireddi RK, Kanneganti TD. The PANoptosome: a deadly protein complex driving pyroptosis, apoptosis, and necroptosis (PANoptosis)[J]. Front Cell Infect Microbiol, 2020, 10: 238. DOI:10.3389/fcimb.2020.00238 |
| [7] | Zhu P, Ke ZR, Chen JX, et al. Advances in mechanism and regulation of PANoptosis: Prospects in disease treatment[J]. Front Immunol, 2023, 14: 1120034. DOI:10.3389/fimmu.2023.1120034 |
| [8] | Yang B, Hu A, Wang TT, et al. SARS-CoV-2 infection induces ZBP1-dependent PANoptosis in bystander cells[J]. Proc Natl Acad Sci USA, 2025, 122(28): e2500208122. DOI:10.1073/pnas.2500208122 |
| [9] | Cui YH, Wang XQ, Lin FY, et al. miR-29a-3p improves acute lung injury by reducing alveolar epithelial cell PANoptosis[J]. Aging Dis, 2022, 13(3): 899. DOI:10.14336/ad.2021.1023 |
| [10] | Xie JL, Fu QJ, Qin LL, et al. Ochratoxin A induces lung cell PANoptosis through activation of the AIM 2 inflammasome[J]. Int Immunopharmacol, 2025, 150: 114184. DOI:10.1016/j.intimp.2025.114184 |
| [11] | Karki R, Sharma BR, Tuladhar S, et al. Synergism of TNF-α and IFN-γ triggers inflammatory cell death, tissue damage, and mortality in SARS-CoV-2 infection and cytokine shock syndromes[J]. Cell, 2021, 184(1): 149-168. e17. DOI:10.1016/j.cell.2020.11.025 |
| [12] | Evrard B, Sinha P, Delucchi K, et al. Causes and attributable fraction of death from ARDS in inflammatory phenotypes of sepsis[J]. Crit Care, 2024, 28(1): 164. DOI:10.1186/s13054-024-04943-x |
| [13] | Du LP, Wang XY, Chen SY, et al. The AIM2 inflammasome: a novel biomarker and target in cardiovascular disease[J]. Pharmacol Res, 2022, 186: 106533. DOI:10.1016/j.phrs.2022.106533 |
| [14] | Sharma BR, Karki R, Kanneganti TD. Role of AIM2 inflammasome in inflammatory diseases, cancer and infection[J]. Eur J Immunol, 2019, 49(11): 1998-2011. DOI:10.1002/eji.201848070 |
| [15] | Liang S, Xing MY, Chen X, et al. Predicting the prognosis in patients with sepsis by a pyroptosis-related gene signature[J]. Front Immunol, 2022, 13: 1110602. DOI:10.3389/fimmu.2022.1110602 |
| [16] | Sagulenko V, Vitak N, Vajjhala PR, et al. Caspase-1 is an apical caspase leading to caspase-3 cleavage in the AIM2 inflammasome response, independent of caspase-8[J]. J Mol Biol, 2018, 430(2): 238-247. DOI:10.1016/j.jmb.2017.10.028 |
| [17] | Li HT, Li Y, Song C, et al. Neutrophil extracellular traps augmented alveolar macrophage pyroptosis via AIM2 inflammasome activation in LPS-induced ALI/ARDS[J]. J Inflamm Res, 2021, 14: 4839-4858. DOI:10.2147/JIR.S321513 |
| [18] | Ao YC, Ma JY, Hou XY, et al. Obesity promotes ARDS by modulating ceramide transfer protein-ceramide pathway and exacerbating oxidative stress/apoptosis in alveolar macrophages[J]. Cell Mol Life Sci, 2025, 82(1): 242. DOI:10.1007/s00018-025-05706-9 |
| [19] | Lee S, Karki R, Wang YQ, et al. AIM2 forms a complex with pyrin and ZBP1 to drive PANoptosis and host defence[J]. Nature, 2021, 597(7876): 415-419. DOI:10.1038/s41586-021-03875-8 |
| [20] | 张晨, 蒲翔, 苏进, 等. 肺泡巨噬细胞诱导免疫应答对肺组织炎性损伤作用机制的研究进展[J]. 基础医学与临床, 2023, 43(10): 1585-1589. DOI:10.16352/j.issn.1001-6325.2023.10.1585 |
| [21] | Wei SW, Wu L, Xiang Z, et al. EIF2AK2 protein targeted activation of AIM2-mediated PANoptosis promotes sepsis-induced acute kidney injury[J]. Ren Fail, 2024, 46(2): 2403649. DOI:10.1080/0886022X.2024.2403649 |
| [22] | 王远扬, 徐昉, 祁海峰. IL-27通过JAK2/STAT1通路促进Th1细胞分化加重急性肺损伤[J]. 重庆医科大学学报, 2024, 49(12): 1572-1576. DOI:10.13406/j.cnki.cyxb.003682 |
| [23] | Ragab D, Salah Eldin H, Taeimah M, et al. The COVID-19 cytokine storm; what we know so far[J]. Front Immunol, 2020, 11: 1446. DOI:10.3389/fimmu.2020.01446 |
| [24] | Damsky W, King BA. JAK inhibitors in dermatology: The promise of a new drug class[J]. J Am Acad Dermatol, 2017, 76(4): 736-744. DOI:10.1016/j.jaad.2016.12.005 |
| [25] | Liu YB, Huang ZC, Wei YL, et al. Identification of STXBP6-IRF1 positive feedback loop in regulation of PD-L1 in cancer[J]. Cancer Immunol Immunother, 2021, 70(2): 275-287. DOI:10.1007/s00262-020-02678-6 |
| [26] | Zenke K, Muroi M, Tanamoto KI. IRF1 supports DNA binding of STAT1 by promoting its phosphorylation[J]. Immunol Cell Biol, 2018, 96(10): 1095-1103. DOI:10.1111/imcb.12185 |
| [27] | Wu JP, Wu C, Ma YJ, et al. AIM2 deficiency alleviates cardiac inflammation and hypertrophy in HFD/STZ-induced diabetic mice by inhibiting the NLRC4/IRF1 signaling pathway[J]. J Cardiovasc Transl Res, 2025, 18(1): 94-109. DOI:10.1007/s12265-024-10556-0 |
| [28] | Liu Q, Xie WL, Wang YT, et al. JAK2/STAT1-mediated HMGB1 translocation increases inflammation and cell death in a ventilator-induced lung injury model[J]. Lab Invest, 2019, 99(12): 1810-1821. DOI:10.1038/s41374-019-0308-8 |
| [29] | Karki R, Kanneganti TD. The 'cytokine storm': molecular mechanisms and therapeutic prospects[J]. Trends Immunol, 2021, 42(8): 681-705. DOI:10.1016/j.it.2021.06.001 |
| [30] | 尉飞, 王湘雨, 刘志勇. 血必净通过miR-155/JAK2/STAT1信号通路对肺炎克雷伯菌所致重症肺炎大鼠肺组织损伤的影响[J]. 实用医学杂志, 2023, 39(18): 2335-2341. DOI:10.3969/j.issn.1006-5725.2023.18.009 |
 2026, Vol. 35
2026, Vol. 35