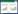脓毒症(sepsis)因具有高致死率成为全球的负担和不断深入研究的热点[1]。脓毒症本质为免疫失衡、炎症级联反应及由之而来的凝血功能紊乱成为导致多器官功能衰竭(MODS)的核心枢纽[2-4]。1991年以感染加SIRS≥2项定义为Sepsis 1,首次建立国际通用标准,并将“感染相关全身炎症”作为核心,解决了此前概念混乱的问题[5]。
显然Sepsis 1缺乏特异性,为改进脓毒症的诊断,Sepsis 2对脓毒症的定义进行了补充,保留Sepsis 1中感染加SIRS,新增多类炎症标记物、器官损伤或功能异常的指标、血流动力学指标,并依据严重程度分为脓毒症、严重脓毒症、感染性休克[6]。Sepsis 2的意义在于细化病情严重程度分层,便于早期干预与预后评估。但是存在指标繁杂、临床难以快速应用、SIRS特异性不足等问题。2016年Sepsis 3的问世[7-9]直接定义为感染导致宿主反应失调引发危及生命的器官功能障碍作为重点。采用SOFA评分如急性升高≥2分诊断脓毒症,取消了敏感性高且特异性低的SIRS概念。将感染性休克归为脓毒症亚型:即需要使用血管加压药物得以维持MAP≥65 mmHg且充分液体复苏后乳酸仍>2 mmol/L。Sepsis 3同时引入了qSOFA快速筛查的建议。其核心涵盖以下几个方面:⑴病理升级,契合病理本质:从“炎症反应”到“宿主反应失调和器官损伤”;⑵诊断精准:SOFA/qSOFA提升特异性,减少非感染性炎症误判,降低老年/免疫低下者漏诊率;⑶休克标准化:明确感染性休克判定,统一临床与科研标准,减少异质性;⑷分层救治:聚焦器官功能,推动早期目标导向治疗,降低病死率[10-15]。2025版sepsis 3.5指南的核心凸显了“快速、精准、预警”。尤其是改良的qSOFA(m-qSOFA)为2025版指南的最大亮点,识别低血压状态而非绝对收缩压数值(SBP≤100 mmHg调整为SBP≤100 mmHg或者较基础值下降≥20 mmHg)和明确“意识状态改变”为避免“烦躁”“嗜睡”等主观描述。上面两项中任何一项阳性均提示为高风险(原Sepsis 3.0定义为低危)[16]。
1.2 脓毒症诱导的凝血病直击脓毒症的病理生理本质 1.2.1 SIC与DIC的区别与弥漫性血管内凝血(disseminated intravascular coagulation, DIC)不同的是,脓毒症诱导的凝血病(sepsis-induced coagulopathy, SIC)的目标人群聚焦感染导致脓毒症或脓毒性休克患者,且SIC概念的提出在疾病病程上早于ISTH的显性DIC概念。DIC的基础病除了脓毒症,还包括创伤、肿瘤、产科急症等诱因。ISTH制定的DIC积分系统是区分二者的核心依据,积分基于血小板计数、凝血酶原时间、纤维蛋白原、纤溶标志物(D-二聚体)四项指标计算,≥5分即可诊断显性DIC;<5分为阴性DIC,需结合临床动态监测,警惕进展为显性DIC。基于SIC定义在不同的团体或者组织赋予的内容存在诸多不同,且DIC项目繁多,不便于操作,为了方便读者记忆,特汇总了SIC评分系统与ISTH显性DIC评分(如表 1所示)及不同医疗组织的SIC评分项特点(如表 2所示)。不难看出只有中国诊断评分系统(Chinese diagnostic scoring system for disseminated intravascular coagulation, CDSS)考虑到了基础病及临床症状[17]。
| 内容 | SIC评分 | ISTH显性DIC评分 |
| 核心理念与目的 | 及早识别已发生凝血系统激活患者及可能向DIC发展中并从干预中获益的可能患者 | 脓毒症晚期的确认及体现已经发生的、广泛的、获得性的凝血功能紊乱综合征的严重程度终极诊断分级 |
| 目标人群 | 脓毒症/脓毒性休克患者 | 所有可能发生DIC的临床情况(如脓毒症、创伤、肿瘤、产科急症等) |
| 前提条件 | 确诊脓毒症(基于SOFA评分≥ 2分) | 存在已知可导致DIC的基础疾病 |
| 评分项目与阈值 | 血小板计数(×109/L) > 150: 0分 100~150: 1分 < 100: 2分 凝血酶原时间-INR < 1.2: 0分 1.2~1.4: 1分 > 1.4: 2分 器官功能衰竭(呼吸、循环、肝脏、肾脏功能)评估 (SOFA) 评分 ≥2: 1分(默认的前提条件) |
血小板计数(×109/L) > 100: 0分 50~100: 1分 < 50: 2分 纤维蛋白降解产物(如D-Dimer) 无升高: 0分 中度升高: 2分 显著升高: 3分 凝血酶原时间延长(s) < 3: 0分 3~6: 1分 > 6: 2分 纤维蛋白原水平(g/L) > 1.0: 0分 ≤1.0: 1分 |
| 计算与诊断阈值 | 0~6分;≥ 4分SIC | 0~8分;≥ 5分为显性DIC |
| 关键项目特点 | 极简、快速、易行:仅含2个凝血实验室指标(血小板、PT/INR),均在急诊/ICU常规检测,数十分钟内可得 | 全面、但耗时:包含4项实验室指标,尤其是纤维蛋白原和FDP/D-Dimer可能非即时可得,耗时更长 |
| 与器官功能关联 | 直接整合SOFA评分作为前提和计分项,将凝血紊乱与器官功能障碍直接挂钩,体现了脓毒症的本质 | 未直接整合,仅评估凝血系统本身的状态,器官功能不全作为结果但未纳入评分体系 |
| 临床行动导向 | 类似于“行动触发器”:诊断为SIC(≥4分)后,立即启动:①更密切的凝血监测;②评估出血/血栓风险;③考虑启动个体化抗凝治疗的决策流程 | 相当于“状态确认书”:诊断为显性DIC后,主要行动是:①确认病情严重性;②启动或加强替代治疗(如输注血小板、凝血因子、纤维蛋白原);③治疗重点更偏向于纠正消耗性凝血病和出血风险 |
| 反映的病理阶段 | 早期/高凝期,凝血系统开始被激活、尚未发生严重消耗的阶段。及时抗凝干预可能最有效 | 中晚期/消耗期,当评分达到诊断标准时,多已发生明显的凝血因子消耗和纤溶系统紊乱,患者可能已处于混合型或低凝/纤溶亢进状态 |
| 时间动态性 | 强调不稳定期每12~24 h动态、连续监测,以追踪病情变化和干预效果。稳定期24~72 h评估 | 常用于某个时间点的诊断确认,动态监测以评估进展 |
| 项目 | 评分 | SIC | ISTH | JAAM[18] | CDSS[19] |
| 基础病 | 2分 | 无 | 无 | 无 | 有 |
| 临床症状 | 1分 | 无 | 无 | 无 | 有 |
| SIRS评分 | 1分 | 无 | 无 | ≥3 | 无 |
| 修正SOFA评分 | 2分 | ≥2 | 无 | 无 | 无 |
| 1分 | ≥1 | 无 | 无 | 无 | |
| 血小板计数 | 3分 | 无 | 无 | <80或24 h内下降50% | |
| 2分 | <100 | <50 | 无 | <80或24h内下降50% | |
| 1分 | ≥100,<150 | ≥50,<100 | ≥80,<120或24 h内下降30%~50% | ≥80,<100 | |
| PT延长 | 2分 | >1.4 | ≥6s | 无 | ≥6 s |
| 1分 | >1.2,≤1.4 | ≥3 s,<6 s | ≥1.2 s | ≥3 s,<6 s或APTT>10 s | |
| FDP(JAAM,μg/mL),D-Dimer(CDSS,mg/L) | 3分 | 明显升高 | ≥25 | ≥9 | |
| 2分 | 无 | 中等程度升高 | 无 | ≥5,<9 | |
| 1分 | 无 | 无 | ≥10,<25 | 无 | |
| 纤维蛋白原(g/L) | 1分 | 无 | <1 | 无 | <1 |
| 诊断 | ≥4 | ≥5 | ≥4 | ≥7 | |
| 注:DIC,disseminated intravascular coagulation,弥漫性血管性内凝血;ISTH,International Society of Thrombosis and Haemostasis,国际血栓与止血学会;JAAM,Japanese Association for Acute Medicine,日本急诊医学会;SIC,Sepsis-Induced Coagulopathy,脓毒症诱导的凝血病;SIRS,Systemic Inflammatory Response Syndrome, 系统性炎症反应综合征;SOFA,Sequential Organ Failure Assessment,序贯器官衰竭评估,CDSS,Chinese diagnostic scoring system中国诊断评分系统 | |||||
需要提醒注意的是,SIC诊断存在一定的局限性:仅包含了两个凝血生物标志物,存在将类似DIC的疾病误诊的风险。如鉴别血栓性微血管病(TMA)相关疾病比较困难。SIC需要与血栓性血小板减少性紫癜(TTP)[20]、非典型溶血性尿毒症综合征(aHUS)[21]以及肝素诱导的血小板减少症(HIT)[22]等鉴别。
1.2.2 SIC病理生理机制SIC核心为脓毒症导致的血管内皮细胞损伤和凝血紊乱,表现为不同程度的凝血系统激活、纤溶失衡及微血栓形成[23-24]。尽管ISTH显性的DIC已经成为公认预测脓毒症高病死率的重要指标[25-26]。当研究逐渐凸显脓毒症导致脏器功能不全的核心为凝血机制紊乱时,研究开始聚焦如何通过恰当地抗凝治疗避免发展到DIC,或者如何找到最佳获益时机成为降低脓毒症患者病死率等焦点问题成为后续对抗凝治疗的关注度不断地升温的重要原因。SIC与DIC虽然均代表凝血机制出现紊乱,但在DIC中,存在明显或严重的消耗性血管内凝血障碍,即血小板、纤维蛋白原和凝血因子显著减少,并伴有血栓形成和(或)出血的临床证据。相比之下,SIC作为一种非显性DIC,被描述为全身性血管内凝血,尚没有明显的血小板、纤维蛋白原和凝血因子消耗[27],并且常常先于DIC的失代偿性凝血病出现。SIC概念的引出最大价值在于提醒临床医生针对脓毒症患者进行早期评估、识别、及时、恰当干预,避免发展到DIC。SIC的实验室特征与真正的DIC分子功能障碍的研究显示存在区别,同时提示SIC定义为内皮病相关的血管微血栓性疾病(endotheliopathy-associated vascular microthrombotic disease, EA-VMTD)更为贴切[28]。2020年之后,脓毒症相关的研究发表的数量呈指数增长趋势。美国遥遥领先(占27.5%),发表在Critical Care Medicine相关文章最多。2001年发表的PROWESS试验为被共同引用次数最多的基础文献[29]。脓毒症与凝血功能障碍研究领域共被引次数较多的作者为日本的Iba及Yamakawa教授。过去二十年中,SIC的研究已从探索基本凝血途径发展到研究免疫血栓形成的复杂机制。研究热点集中在内皮损伤、NETs以及精确诊断和治疗策略的发展上。尤其是固有免疫和凝血系统之间的非常复杂的病理生理变化[30]。现有脓毒症的研究,主要集中在以下四个方面:病理生理机制与内皮损伤;生物标志物与预后评估;抗凝治疗与临床管理策略;免疫血栓形成与中性粒细胞胞外捕网(NETs)。本文重点聚焦SIC的病理生理机制(如图 1所示)。
|
| 图 1 SIC的病理生理机制 |
|
|
研究发现脓毒症通过微生物相关的分子模式(pathogen-associated molecular patterns, PAMPs)或损伤相关分子模式(damage-associated molecular patterns, DAMPs)表达并进一步激活免疫细胞,并促使单核细胞、内皮细胞和中性粒细胞表达转录因子,进而引发自身调控极强的正反馈信号分子——凝血酶的生成。活化的NETs为多种阴离子提供必要的平台,进一步促进凝血酶生成[31-32]。血小板脱颗粒、白细胞及组织因子表达、NETs形成以及血小板-白细胞聚集体形成促进凝血酶生成,进而导致纤维蛋白产生和血小板消耗,最终引发微血管血栓形成[33-34]。
脓毒症因存在广泛的细胞损伤释放DAMPs,并通过特定的Toll样受体(TLRs)发出与初始感染促炎或促凝反应信号无关。其中组蛋白和高迁移率族蛋白B1(HMGB-1)通过细胞表面的Toll样受体2/4(TLR2/4)启动,循环游离DNA(cfDNA)、单链RNA(ssRNA)和双链RNA(dsRNA)分别通过内体中的Toll样受体9(TLR9)、Toll样受体7/8(TLR7/8)和Toll样受体3(TLR3)启动。上述受体的激活会导致转录因子NF-κB和AP-1通过MyD88进行核转位,但TLR3信号通路除外,该信号诱导Trif信号传导和IRF的转位。上述信号通路会进一步导致免疫细胞中促炎细胞因子和TF表达的诱导[35-37]。
促炎及抗炎因素的异常表达进一步如TNF-a、IFNs、白介素如IL-6, IL-2、IL-12、IL-4、IL-10及转移生长因子(TGF-β) 等均参与了凝血病的发展过程[38-39]。研究发现,补体同样参与了SIC炎症激活的过程,如C1q、C3、C3a、C4a、C4BP、C5、C5a、C5aR、C5b - 9、MASP和FI参与内皮细胞、血小板激活的整个过程[40]。
1.2.2.2 内皮病变加重血管内微环境的失衡完整的内皮细胞(ECs)可调节血浆抗凝蛋白水平,包括抗凝血酶(AT)、组织因子途径抑制物(TFPI)和蛋白C[41-42]。ECs具有应对剪切应力以及适应血液供应等特点,能够有效地防止在血流动力学不稳定时各种功能细胞的不适当黏附和激活。完好的内皮糖萼通过释放多种抗血栓物质和表达抗凝血因子对血管止血调节中发挥重要的作用[12, 43-44]。ECs虽非免疫细胞,但可以通过各种表型和功能的修饰参与对外部病原体和内毒素以及随后的细胞因子风暴的应答。ECs功能紊乱会加剧炎症和凝血功能障碍,尤其在脓毒症的早期阶段因导致血管通透性增加,并通过免疫性血栓形成参与血管渗漏和微循环障碍[45]。肝素酶暴露了覆盖着糖萼的含有凝血因子(FⅡ、FⅦ、FⅨ和FⅩ)磷脂酰丝氨酸,并诱导血管内凝血形成[41, 46]。
受损的ECs分泌Weibel-Palade小体,增加循环中黏附分子(如vWF和P选择素)的水平,参与促进血小板和白细胞的活化、黏附、聚集以及内皮表型转化[46]。研究同时发现内皮细胞胞吐过程中超大von Willebrand因子(ULVWF)的形成,被认为是微血栓形成的触发起始因素[28, 47]。脓毒症患者存在消耗增加、合成减少、蛋白水解清除以及针对金属蛋白酶抑制物的存在,ADAMTS13对ULVWF多聚体的快速蛋白水解受到限制[48]。脓毒症患者的非酶/酶抗氧化系统消除氧化应激的等效性缺失,在微循环功能障碍时会进一步加重导致促凝血状态[49-52]
1.2.2.3 天然抗凝物质减少完整的ECs可防止抗凝血酶水平下降,并产生抗炎作用[53-54]。脓毒症病情进展会导致机体天然抗凝物质如抗凝血酶(AT)、蛋白C和组织因子途径抑制物(tissue factor pathway inhibitor, TFPI)减少,在确保高效的抗凝和抗炎机制方面的作用降低[41, 55]。抗凝血酶(AT)作为重要的防止内皮糖萼脱落的天然抗凝物质在脓毒症期间显著减少[56-58]。且抗凝血酶活性(理想情况下为80%,至少70%以上)与疾病严重程度和生存率高度相关[59-60]。蛋白C、蛋白S、血栓调节蛋白(TM)和活化蛋白C(APC)作为ECs的保护因子,通过多种机制如刺激ECs上的蛋白酶激活受体(PAR),以及裂解细胞毒性组蛋白对抗炎症相关的内环境紊乱[61-62]。研究发现脓毒症期间,蛋白C和TM水平下调[58, 63-64]。TFPI通过抑制FVII激活蛋白酶和因子Xa,以及通过Xa-TFPI复合物形成来抑制FVIIa-TF复合物[65]。
脓毒症早期,过度生成的纤溶酶可介导TFPI的裂解和失活[66]。TFPI生成失衡无法使由激活的免疫细胞和内皮细胞大量表达组织因子(TF)所触发的凝血级联反应失活。激活的中性粒细胞释放的FXa、凝血酶、中性粒细胞弹性蛋白酶(NE)和髓过氧化物酶(MPO)均可氧化TFPI,使之活性下降。且脓毒症患者血浆凝血酶激活纤溶抑制物(thrombin activatable fibrinolysis inhibitor, TAFI) 和TFPI因细菌类型不同存在不同程度的变化[67]。
1.2.2.4 凝血/纤维蛋白溶解失衡尽管纤维蛋白降解产物在器官功能障碍患者中显著升高,但在脓毒症诱导DIC中却显著下降[68-70]。α2-抗纤溶酶(A2AP)作为体内血浆纤溶酶(Pla)的主要抑制物,在剪切应力增加的条件下,A2AP与纤维蛋白网状结构的交联变得更加高效。因此,ECs表面超大von Willebrand因子(ULVWF)的存在起到了相对的保护作用[71]。纤溶酶原激活物抑制物-1(PAI-1)的血清水平与脓毒症患者循环中白细胞介素-6(IL-6)的水平呈正相关,尤其发生在合并器官衰竭和DIC的患者中[72-74],并受肿瘤坏死因子-α(TNF-α)驱动[75]。NETs形成过程中会释放DNA-组蛋白复合物,并通过与组织型纤溶酶原激活剂(t-PA)和尿激酶型纤溶酶原激活剂(uPA)相互作用延长纤溶作用[76]。研究表明,NETs可作为内部介质,通过增强纤维骨架支撑形成更粗的纤维蛋白纤维,增加血栓的异质性[12, 76]。同时NETs产生的碎片会抑制微循环中的纤溶作用,使脓毒症期间的微血栓对抗凝剂更具抵抗性[63, 77]。凝血酶激活的纤溶抑制物(TAFI)激活同时影响凝血酶生成和血栓稳定性,并通常导致富含血小板的血栓出现纤溶抵抗。
1.2.2.5 免疫血栓与血栓炎症交互影响病原体入侵并被感染部位识别后,机体恰当的反应为局部免疫血栓形成(血管内血栓形成的过程)[78],既有助于清除病原体,又不会引发固有免疫反应的弥漫性激活[79-80]。脓毒症中的免疫血栓形成完全是基于炎症和补体系统的一系列激活引发,失控的多种细胞因子的释放导致免疫细胞和非免疫细胞表面蛋白表达发生改变,进而对炎症事件产生反馈和前馈效应,上述现象被称为血栓炎症[81-82]。研究进一步证明免疫血栓形成和血栓炎症在脓毒症期间会加剧血管内损伤和全身性损害[52, 78, 83-85]。脓毒症患者中凝血酶在与炎症和凝血相关的多种通路中发挥关键作用,通过激活PARs(包括PAR-1、PAR-3和PAR-4)导致屏障破坏,进而诱导M1型巨噬细胞极化,直接激活ECs,并进一步促进了NF-κB通路的激活[86-88]。凝血酶具有上调内皮细胞、平滑肌细胞、成纤维细胞、上皮细胞和单核细胞中的IL-1、IL-6、IL-8、TNF-α、TGF-β、ICAM-1、VCAM-1和P选择素[89-91],激活血栓形成时还能诱导MCP-1、VEGF、金属蛋白酶和PDGF的释放,进一步促进炎症[92-95]。
2 SIC概念引出的重要价值及其相关研究 2.1 PROWESS-SHOCK研究成为SIC抗凝治疗的重要铺垫PROWESS-SHOCK团队研究[96-98]旨在通过前瞻性随机对照试验观察重组人活化蛋白在脓毒性休克患者的安全性及有效性研究。该研究主要涉及六个层面:脓毒性休克、肺炎导致的急性呼吸窘迫综合征、腹部手术后、严重的脓毒症、肺炎/急性呼吸窘迫综合征/多脏器功能衰竭、迟发脓毒性休克[99]。该研究证实重组人活化蛋白抗凝并非使所有严重脓毒症或脓毒性休克患者获益,无选择性使用并没有改善生存率,反而增加出血风险。PROWESS试验的亚组分析表明,与给予活化蛋白C相关的生存获益在基线时往往会随着疾病严重程度的增加而增加[100]。此外,对KyberSept试验进行的post hoc亚组分析,结果显示抗凝血酶治疗可能会改善简化APACHEⅡ预计病死率为30%~60%的患者生存率[101]。一项以评估重组血栓调节蛋白(rTM)治疗的生存效应与对照病死率之间的关系Meta分析显示,随着对照组死亡风险的增加,rTM治疗有益的概率显著增加[102-103]。
很显然,PROWESS-SHOCK从反向论证了“精准抗凝”必要性,更为后续的亚组分析和新的研究做出了重要的铺垫——揭示了未来寻找可能从抗凝治疗中获益的脓毒症患者亚群的必要性。
2.2 Iba及Yamakawa团队为寻找SIC抗凝时机拉开序幕 2.2.1 Iba团队研究及发现2017年Iba教授团队对日本全国数据库进行的大型回顾性队列研究证实SIC诊断标准的预后价值[104]。该研究汇总1 498例接受rTM治疗的患有脓毒症和凝血病并发症的日本患者。该研究终点:在开始治疗前即刻测得的血小板计数、凝血酶原时间(PT)比值、纤维蛋白原/纤维蛋白降解产物、全身炎症反应综合征评分和序贯器官衰竭评估(SOFA)评分与28 d病死率相关性进行了检查。逻辑回归结果显示血小板计数、PT比值和总SOFA评分为死亡结局的独立预测因素。SIC阳性阈值(≥4分)对预测28 d病死率具有较高的预测价值。与当时JAAM-DIC发布的DIC评分相比,SIC评分预测的28 d病死率更高(38.4% vs. 34.7%)。为评估SIC评分是否可指导启动抗凝治疗的决策,研究将SIC明确定义为DIC的早期阶段,并创建了包含血小板计数、PT/INR和序贯器官衰竭评估评分的简易诊断评分系统,更为早期识别和干预提供了工具。所以,该研究为“SIC评分≥4分可作为启动抗凝的筛选工具”提供了最直接的临床证据。Iba团队的研究明确SIC向显性DIC进展的病理生理机制,并提出由SIC向显性DIC进展机制的“两步法”诊断流程(先筛SIC,再评估显性DIC),以优化临床决策[105]。该策略在同年ISTH官方诊断路径共识中推出,正式推荐SIC作为DIC早期识别工具,形成“SIC筛查→显性DIC确诊”的标准化路径,推动全球临床应用[106]。Iba团队的回顾性研究仍需前瞻性研究证实SIC评分≥4分患者接受抗凝治疗的有效性和安全性。于是,SIC作为DIC的早期预警指标评分,与器官功能障碍(SOFA评分)和28 d病死率强相关研究陆续被多项研究证实,并成为临床试验和潜在治疗干预的筛选工具,进一步验证SIC评分≥4分可早于ISTH显性DIC,且能预测28 d死亡与显性DIC进展[107]。
随后Iba团队研究发现,SIC评分≥4分患者中,rTM可降低显性DIC进展率与28 d病死率,且出血风险可控。进一步验证血小板计数、PT比值、SOFA评分等标志物对SIC早期诊断的价值,为临床快速识别提供依据[24, 108-109]。
2.2.2 Yamakawa团队聚焦SIC的转化研究Yamakawa团队核心方向侧重SIC抗凝疗效的验证、COVID - 19相关凝血病对比、SIC分层治疗与真实世界的研究。曾探索早期重组人可溶性血栓调节蛋白(rhTM)疗效探索并验证rhTM在SIC患者的生存获益,为后续SIC抗凝提供早期依据[110]。随后通过多中心倾向评分分析显示rhTM可降低脓毒症DIC患者病死率,且出血风险可控,明确SIC高危人群获益规律及脓毒症DIC中rhTM获益特征[103]。进一步研究发现SIC评分4~5分的患者,早期普通肝素可降低住院病死率,且不增加严重出血风险,为中度SIC抗凝提供真实世界证据[111]。
Yamakawa团队早期大样本队列来自日本42家ICU的全国多中心回顾性注册研究,纳入2663例脓毒症患者,揭示SIC/DIC与SOFA分层下抗凝治疗的生存获益特征,奠定SIC抗凝获益基础[112]。随后通过日本全国重症监护数据库的一项回顾性、多中心队列研究,纳入超过28 000名符合Sepsis-3标准的成年患者,比较了在确诊脓毒症后3 d内接受治疗剂量肝素抗凝的患者与未接受抗凝的患者之间的病死率差异。研究采用倾向性评分匹配来平衡组间差异。结果显示,对于SIC评分≥4分的患者,早期接受肝素抗凝与28 d全因病死率显著降低相关。而对于SIC评分<4分的患者,抗凝则未显示出明确的生存获益。该研究的重要贡献在于是“精准抗凝”理念在脓毒症领域的重要实践,首次通过大规模人群研究,为“SIC评分≥4分成为筛选可能从抗凝治疗中获益患者的重要工具”这一观点提供了强有力的临床数据支持。验证SIC评分对识别抗凝治疗获益人群的价值,证实SIC评分≥4分患者抗凝获益更显著,实现了对SIC评分抗凝价值进行了外部验证[113]。Yamakawa团队研究部分再次证实了PROWESS-SHOCK研究没有发现抗凝治疗获益的局限性在于没有对患者进行精准分层。但是,Yamakawa团队研究局限于回顾性观察性研究,抗凝治疗的最终决策仍需结合出血风险、器官功能等临床因素综合判断。
2024年Yamakawa团队的一项Meta研究未看到肝素的益处[114]。该研究在1 733篇初始文章中,只有3项研究符合纳入标准。该分析纳入了426名患者,结果显示肝素组和对照组在28 d病死率和住院病死率差异无统计学意义(RR=0.86,95%CI:0.60~1.24)。对脓毒症相关弥散性血管内凝血(DIC)患者(n = 109)的亚组分析也未显示病死率有显著降低(RR= 0.84,95%CI :0.51~1.38)。异质性为零,且未观察到发表偏倚。此外,出血并发症差异有统计学意义(RR=0.49,95%CI 0.24~0.99,P=0.047)。最新的《脓毒症性凝血病诊疗中国专家共识2024》也提到,SIC评分为抗凝治疗时机的选择提供了依据[17]。
3 SIC识别及抗凝治疗时机目前关于抗凝治疗对脓毒症患者临床益处的数据不一,拯救脓毒症运动尚不建议进行针对性的抗栓治疗[115]。同时,凝血功能障碍的诊断标准不同和(或)不论凝血状态将所有脓毒症患者广泛纳入研究,可能导致了临床试验结果不一致。抗凝治疗带来的更好疗效可能仅在一部分脓毒症患者中得到证实。在其他评估针对SIC和(或)DIC的抗凝治疗的临床研究中,报道了体内凝血酶生成减少及凝血参数改善的证据[18, 116-117]。
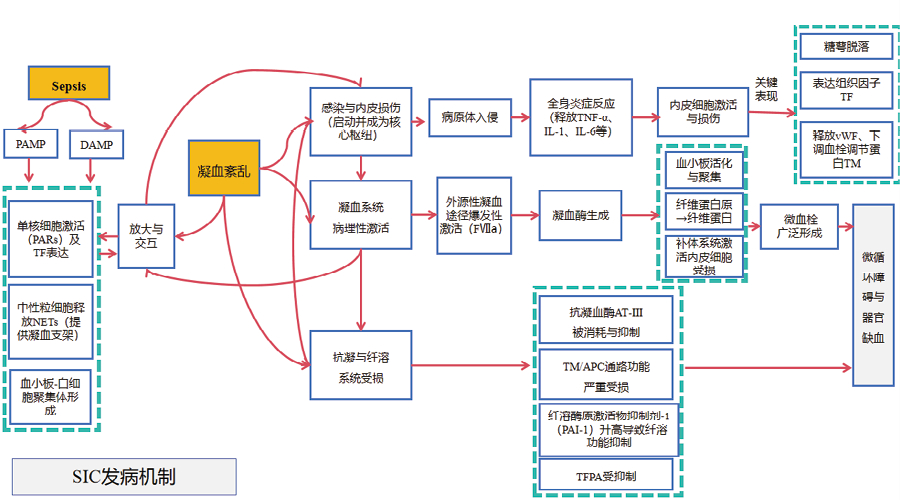
3.1 脓毒症患者管理及SIC识别Tejada等研究发现通过监测循环中的NETs有助于早期识别急诊脓毒症患者的栓塞风险[118],该研究回顾212例在急诊科就诊时被诊断为脓毒症的成年患者。通过酶联免疫吸附测定法检测血浆NETs、白细胞介素-6(IL-6)和中段前肾上腺髓质素(MR-ProADM)。研究发现急诊就诊时对NETs进行定量分析,可能有助于识别脓毒症中高风险的免疫血栓形成表型,并在抗凝治疗的同时支持更早开展针对NETs的靶向治疗。脓毒症患者的管理从诊断开始,需要针对SIC进行第一时间且反复评估,SIC是否需要抗凝治疗需要“四步法”决策,见图 2。
|
| 图 2 SIC抗凝决策“四步法” |
|
|
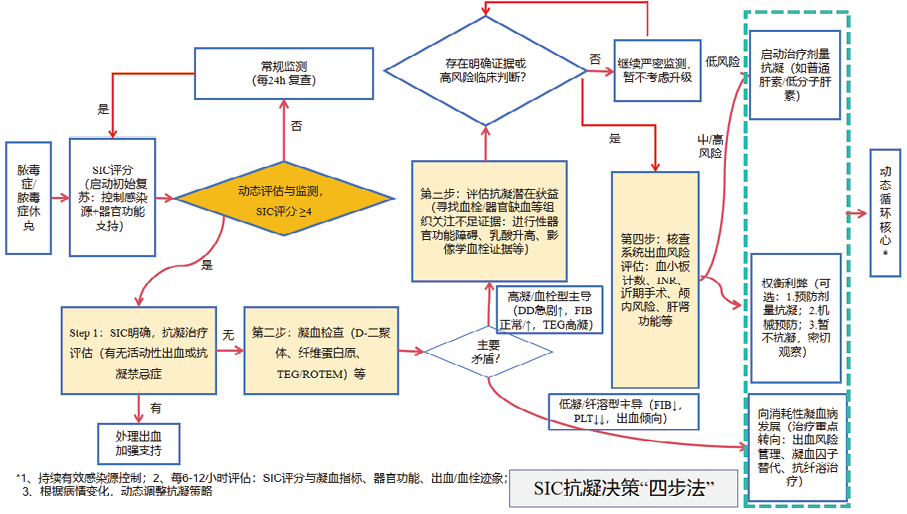
抗凝治疗在抗感染的同时可以有效控制DIC病情进展[84]。而SIC超前于DIC,抗凝治疗时机的掌握需要更加精准以避免不必要的出血时间或者通过SIC寻找/筛选出抗凝治疗策略。为了方便理解,特汇总为下面流程图,见图 3。
|
| 图 3 脓毒症患者抗凝筛选流程图 |
|
|
肝素在sepsis治疗中的作用远远超过单纯抗凝作用[119]。随着研究深入发现,SIC患者的抗凝治疗兼顾了凝血紊乱和非抗凝治疗的双重作用[120]。肝素具有抗炎、免疫调节和抗补体活性等相互依赖且相互作用的生物学特性,除抗凝血作用外。低分子量肝素(LMWH)的调节作用比普通肝素(UFH)更为突出。尽管目前研究的治疗效果仍存在不一致之处[58, 83, 121-123]。脓毒症抗凝治疗期间,肝素可选择性地与细胞黏附、迁移、增殖和分化的关键调节因子结合,并改变血栓炎症的病理进程,从而改善全身微循环[124]。肝素可中和细胞因子和趋化因子,减少NETs,并抑制放大促炎因子的反馈环路[58]。肝素对带正电荷的蛋白质具有高亲和力,能与免疫血栓形成过程中的关键效应分子选择性作用,尤其是硫酸乙酰肝素结合蛋白[76, 125]。循环细胞外组蛋白介导与脓毒症患者死亡相关的细胞毒性和促凝血活性[123, 126]。由于其结构与肝素硫酸(HS)相似,肝素能够中和内皮糖萼脱落释放的碎片[120, 127]。肝素功能化吸附剂通过去除免疫性血栓形成的核心效应因素,在减少血栓并发症方面显示出相似的有效性,其作用独立于AT及其与肝素抗凝血酶结合戊糖的相互作用[125, 128-129]。体外动物实验表明,经过纯化且去除了抗凝血作用但保留其他独立机制的肝素,能够缓解血栓性炎症和SIC(血栓性炎症相关凝血病)的进展,同时降低出血风险[123, 128]。因此,肝素凭借其抗凝血作用和非抗凝血作用,在脓毒症及脓毒症相关凝血病的治疗中展现出巨大潜力。
APC为一种天然的介导蛋白,通过切割细胞毒性组蛋白H3以及减轻脓毒症诱导的血小板、中性粒细胞与内皮细胞之间的相互作用来调节多种病理状态[130]。重组人活化蛋白C(rhAPC)为首个应用于高病死率脓毒症患者中的APC变体,于2001年获得美国FDA批准[131-133]。尤其是在脓毒症患者中,由于其凝血系统已受到广泛干扰,进一步的多器官并发症(常见为出血相关不良事件)极易被诱发[131, 133-135]。在严重脓毒症且死亡风险较低(APACHEⅡ评分<25或单一器官受累)的患者中,静脉输注rhAPC[剂量为24 μg/(kg·h),持续96 h]未显示出临床获益,但显著增加了出血事件的风险[136]。因此,由于其在脓毒症亚表型中临床获益存在差异以及可能增加的出血风险,rhAPC于2011年从抗凝药物市场撤市[132, 134, 137-138]。
血小板减少症与脓毒症的不良预后相关。抗血小板策略旨在通过降低血小板反应性来减缓凝血病的进展,血小板在介导免疫血栓形成与血栓炎症之间发挥重要作用。抗血小板策略旨在预防广泛的微血栓形成及进展为DIC[139-141]。抗血小板药物可降低炎症生物标志物(如C反应蛋白和P选择素),并抑制白细胞-血小板聚集[142-144]。近年来,阿司匹林(ASA)在脓毒症患者中的应用被认为具有抗炎活性和细菌控制作用。一项纳入689 897例脓毒症患者的荟萃分析显示,ASA能有效降低病死率并改善预后[145-146]。尤其是在管理无明确心血管问题的脓毒症患者时,ASA在降低病死率方面效果更强[147-148],同时也发现对于存在心血管疾病的脓毒症患者,ASA仍能显示脓毒症患者90 d病死率有所降低[149]。
3.3.2 SIC抗凝药物选择2024年中国脓毒血症专家共识建议在存在适应证时建议使用的抗凝药物, 见表 3。
| 药物分类 | 半衰期 | 代谢途径 | 初始剂量 | 给药方式 | 监测方法 | 并发症 | 推荐意见 |
| 肝素类 | |||||||
| 普通肝素 | 60~90 min | 肾脏 | 1~4 U/(kg·h) | 静脉注射 | TEG肝素酶对比试验:R普通检测/R肝素酶检测的比价为1.5~2.0;抗Xa活性0.5~0.7 U/mL;(3)APTT延长至基线值的1.5倍以内 | 出血、肝素诱导的血小板减少症 | 推荐 |
| 低分子肝素 | 2.0~4.5 h | 肾脏 | 50 U/kg,1次/12 h | 皮下或静脉注射 | (1)皮下注射后4 h或静脉注射后10 min的抗Xa活性为0.3~0.5 U/ml;(2)动态监测抗Xa活性或TEG-R时间 | 出血、肝素诱导的血小板减少症 | 推荐 |
| 磺达肝素钠 | 17~21 h;老年人和肾功能不全者延长 | 肾脏 | 1.5~2.5 mg,1次/d | 皮下注射 | 抗Xa活性或TEG-R时间 | 出血、恶心、呕吐、肝酶升高 | 不推荐 |
| 丝氨酸蛋白酶抑制物 | |||||||
| 甲磺酸萘莫司他 | 5~8 min | 80%经肝脏,20%由红细胞降解 | 2~20 mg/h | 静脉注射 | APTT或TEG-R时间 | 高钾血症、出血 | 推荐 |
| 直接凝血酶抑制物 | |||||||
| 阿加曲班 | 39~51 min | 肝脏代谢 | 0.2 μg/(kg·min) | 静脉注射 | APTT或TEG-R时间至基线值的1.5倍以内 | 出血、肝功能异常 | 不推荐 |
| 比伐卢定 | 25 min | 80%蛋白水解,20%经肝脏 | 0.8 μg/(kg·min) | 静脉注射 | APTT或TEG-R时间至基线值的1.5倍以内 | 疼痛、低血压、恶心 | 不推荐 |
| 注:SIC为脓毒症性凝血病;TEG为血栓弹力图;APTT为活化部分凝血活酶时间 | |||||||
SIC可以视为传统DIC诊断标准背景下的“精准化”和“临床导向”的革新。SIC概念的意义,在于为临床医生提供了一个从“该不该抗凝”的模糊争论,转向“如何为这些特定患者评估抗凝指征”的清晰框架。SIC评分(≥ 4分)提示凝血系统已病理性激活,但患者可能处于以凝血激活、微血栓形成为主的早期/高凝期(早期高凝状态是抗凝治疗可能获益最大的窗口期)或者凝血因子和血小板大量消耗的晚期/纤溶亢进期(纤溶系统过度激活甚至为严重出血倾向,此期抗凝治疗风险极高,往往会因为担心出血不建议常规使用,需要动态评估患者的临床表型及抗凝治疗的风险获益)。面对异质性极强的SIC患者,纤溶亢进期是否获益于理论上对减少凝血因子的进一步消耗的益处,其时机仍需要进一步研究及考证。
SIC接受抗凝治疗尚未形成统一标准,但方向明确。仍存在未解问题,如最佳抗凝药物选择(肝素、直接Xa因子抑制物)、确切治疗窗口、不同病原体感染的差异;抗凝以外的靶点如NETs抑制物、内皮保护剂(rTM、可溶性血管生成素-2抗体)、补体抑制物等。仍需要研究床旁即时监测技术如血栓弹力图/旋转式血栓弹力测定在实时指导抗凝中的潜在价值;仍需要更具特异性的生物标志物(如凝血酶-抗凝血酶复合物、可溶性血栓调节蛋白、组织纤溶酶原激活物-抑制物复合物等),以探讨在鉴别凝血表型及鉴定抗凝获益中的作用。
总之,SIC概念的提出,标志着当前管理脓毒症的核心虽然是感染源控制与器官支持,但是脓毒症凝血管理从“晚期、笼统诊断”转向“早期、精准预警”。为在正确的时间、正确的患者及选择正确的抗凝治疗提供了科学框架。
利益冲突 所有作者声明无利益冲突
| [1] | Rudd KE, Johnson SC, Agesa KM, et al. Global, regional, and national sepsis incidence and mortality, 1990-2017: analysis for the Global Burden of Disease Study[J]. Lancet, 2020, 395(10219): 200-211. DOI:10.1016/S0140-6736(19)32989-7 |
| [2] | Lorencio Cardenas C, Yebenes JC, Vela E, et al. Trends in mortality in septic patients according to the different organ failure during 15 years[J]. Crit Care, 2022, 26(1): 302. DOI:10.1186/s13054-022-04176-w |
| [3] | Schmoch T, Mohnle P, Weigand MA, et al. The prevalence of sepsis-induced coagulopathy in patients with sepsis - a secondary analysis of two German multicenter randomized controlled trials[J]. Ann Intensive Care, 2023, 13(1): 3. DOI:10.1186/s13613-022-01093-7 |
| [4] | Tanaka C, Tagami T, Kudo S, et al. Validation of sepsis-induced coagulopathy score in critically ill patients with septic shock: post hoc analysis of a nationwide multicenter observational study in Japan[J]. Int J Hematol, 2021, 114(2): 164-171. DOI:10.1007/s12185-021-03152-4 |
| [5] | Bone RC, Balk RA, Cerra FB, et al. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine[J]. Chest, 1992, 101(6): 1644-1655. DOI:10.1378/chest.101.6.1644 |
| [6] | Dellinger RP, Carlet JM, Masur H, et al. Surviving Sepsis Campaign guidelines for management of severe sepsis and septic shock[J]. Crit Care Med, 2004, 32(3): 858-873. DOI:10.1097/01.ccm.0000117317.18092.e4 |
| [7] | Singer M, Deutschman CS, Seymour CW, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3)[J]. JAMA, 2016, 315(8): 801-810. DOI:10.1001/jama.2016.0287 |
| [8] | Singer M. The new sepsis consensus definitions (Sepsis-3): the good, the not-so-bad, and the actually-quite-pretty[J]. Intensive Care Med, 2016, 42(12): 2027-2029. DOI:10.1007/s00134-016-4600-4 |
| [9] | Timsit JF, Perner A. Sepsis: find me, manage me, and stop me![J]. Intensive Care Med, 2016, 42(12): 1851-1853. DOI:10.1007/s00134-016-4603-1 |
| [10] | Girardis M, David S, Ferrer R, et al. Understanding, assessing and treating immune, endothelial and haemostasis dysfunctions in bacterial sepsis[J]. Intensive Care Med, 2024, 50(10): 1580-1592. DOI:10.1007/s00134-024-07586-2 |
| [11] | Almskog LM, Agren A. Thromboinflammation vs. immunothrombosis: strategies for overcoming anticoagulant resistance in COVID-19 and other hyperinflammatory diseases. Is ROTEM helpful or not?[J]. Front Immunol, 2025, 16: 1599639. DOI:10.3389/fimmu.2025.1599639 |
| [12] | Maneta E, Aivalioti E, Tual-Chalot S, et al. Endothelial dysfunction and immunothrombosis in sepsis[J]. Front Immunol, 2023, 14: 1144229. DOI:10.3389/fimmu.2023.1144229 |
| [13] | Wu M, Yan Y, Xie X, et al. Effect of endothelial responses on sepsis-associated organ dysfunction[J]. Chin Med J (Engl), 2024, 137(23): 2782-2792. DOI:10.1097/CM9.0000000000003342 |
| [14] | Pieterse E, Rother N, Garsen M, et al. Neutrophil extracellular traps drive endothelial-to-mesenchymal transition[J]. Arterioscler Thromb Vasc Biol, 2017, 37(7): 1371-1379. DOI:10.1161/ATVBAHA.117.309002 |
| [15] | Iba T, Levy JH. Inflammation and thrombosis: roles of neutrophils, platelets and endothelial cells and their interactions in thrombus formation during sepsis[J]. J Thromb Haemost, 2018, 16(2): 231-241. DOI:10.1111/jth.13911 |
| [16] | Pisano A, Venditto M, Palmieri C, et al. Novel therapies and interventions in sepsis and septic shock[J]. BJA Educ, 2025, 25(5): 206-217. DOI:10.1016/j.bjae.2025.01.003 |
| [17] | Wei Q, Wang M, Peng X, et al. Comparison of three different disseminated intravascular coagulation (DIC) criteria and diagnostic and prognostic value of antithrombin investigation in patients with confirmed sepsis-induced coagulopathy (SIC)[J]. Clin Appl Thromb Hemost, 2024, 30: 10760296241271334. DOI:10.1177/10760296241271334 |
| [18] | Gando S, Iba T, Eguchi Y, et al. A multicenter, prospective validation of disseminated intravascular coagulation diagnostic criteria for critically ill patients: comparing current criteria[J]. Crit Care Med, 2006, 34(3): 625-631. DOI:10.1097/01.ccm.0000202209.42491.38 |
| [19] | Wang M, Kou H, Deng J, et al. Retrospective evaluation of new chinese diagnostic scoring system for disseminated intravascular coagulation[J]. PLoS One, 2015, 10(6): e0129170. DOI:10.1371/journal.pone.0129170 |
| [20] | Habe K, Wada H, Ito-Habe N, et al. Plasma ADAMTS13, von Willebrand factor (VWF) and VWF propeptide profiles in patients with DIC and related diseases[J]. Thromb Res, 2012, 129(5): 598-602. DOI:10.1016/j.thromres.2011.10.011 |
| [21] | Laurence J, Haller H, Mannucci Pm, et al. Atypical hemolytic uremic syndrome (aHUS): essential aspects of an accurate diagnosis[J]. Clin Adv Hematol Oncol, 2016, 14 Suppl 11(11): 2-15. |
| [22] | Greinacher A, Selleng K, Warkentin TE. Autoimmune heparin-induced thrombocytopenia[J]. J Thromb Haemost, 2017, 15(11): 2099-2114. DOI:10.1111/jth.13813 |
| [23] | Tsantes AG, Parastatidou S, Tsantes EA, et al. Sepsis-induced coagulopathy: an update on pathophysiology, biomarkers, and current guidelines[J]. Life (Basel), 2023, 13(2). DOI:10.3390/life13020350 |
| [24] | Iba T, Umemura Y, Wada H, et al. Roles of coagulation abnormalities and microthrombosis in sepsis: pathophysiology, diagnosis, and treatment[J]. Arch Med Res, 2021, 52(8): 788-797. DOI:10.1016/j.arcmed.2021.07.003 |
| [25] | Wada H, Matsumoto T, Y Y. Diagnosis and treatment of disseminated intravascular coagulation (DIC) according to four DIC guidelines[J]. J Intensive Care, 2014, 2(1): 15-23. DOI:10.1186/2052-0492-2-15 |
| [26] | Gando S, Levi M, Toh CH. Disseminated intravascular coagulation[J]. Nat Rev Dis Primers, 2016, 2: 16037. DOI:10.1038/nrdp.2016.37 |
| [27] | Nickel KF, Renne T. Crosstalk of the plasma contact system with bacteria[J]. Thromb Res, 2012, 130 Suppl 1: S78-83. DOI:10.1016/j.thromres.2012.08.284 |
| [28] | Chang JC. Sepsis and septic shock: endothelial molecular pathogenesis associated with vascular microthrombotic disease[J]. Thromb J, 2019, 17: 10. DOI:10.1186/s12959-019-0198-4 |
| [29] | Bernard Gr, Vincent Jl, Laterre Pf, et al. Efficacy and safety of recombinant human activated protein C for severe sepsis[J]. N Engl J Med, 2001, 344(10): 699-709. DOI:10.1056/NEJM200103083441001 |
| [30] | Man C, An Y, Wang GX, et al. Recent advances in pathogenesis and anticoagulation treatment of sepsis-induced coagulopathy[J]. J Inflamm Res, 2025, 18: 737-750. DOI:10.2147/JIR.S495223 |
| [31] | Fuchs TA, Brill A, Duerschmied D, et al. Extracellular DNA traps promote thrombosis[J]. Proc Natl Acad Sci USA, 2010, 107(36): 15880-15885. DOI:10.1073/pnas.1005743107 |
| [32] | Martinod K, Demers M, Fuchs TA, et al. Neutrophil histone modification by peptidylarginine deiminase 4 is critical for deep vein thrombosis in mice[J]. Proc Natl Acad Sci U S A, 2013, 110(21): 8674-8679. DOI:10.1073/pnas.1301059110 |
| [33] | Mcdonald B, Davis RP, Kim SJ, et al. Platelets and neutrophil extracellular traps collaborate to promote intravascular coagulation during sepsis in mice[J]. Blood, 2017, 129(10): 1357-1367. DOI:10.1182/blood-2016-09-741298 |
| [34] | Khakpour S, Wilhelmsen K, Hellman J. Vascular endothelial cell Toll-like receptor pathways in sepsis[J]. Innate Immun, 2015, 21(8): 827-846. DOI:10.1177/1753425915606525 |
| [35] | Chen F, Zou L, Williams B, et al. Targeting toll-like receptors in sepsis: from bench to clinical trials[J]. Antioxid Redox Signal, 2021, 35(15): 1324-1339. DOI:10.1089/ars.2021.0005 |
| [36] | Clark SR, Ma AC, Tavener SA, et al. Platelet TLR4 activates neutrophil extracellular traps to ensnare bacteria in septic blood[J]. Nat Med, 2007, 13(4): 463-469. DOI:10.1038/nm1565 |
| [37] | Shin HS, Xu F, Bagchi A, et al. Bacterial lipoprotein TLR2 agonists broadly modulate endothelial function and coagulation pathways in vitro and in vivo[J]. J Immunol, 2011, 186(2): 1119-1130. DOI:10.4049/jimmunol.1001647 |
| [38] | Chousterman BG, Swirski FK, Weber GF. Cytokine storm and sepsis disease pathogenesis[J]. Semin Immunopathol, 2017, 39(5): 517-528. DOI:10.1007/s00281-017-0639-8 |
| [39] | Sikora JP, Karawani J, Sobczak J. Neutrophils and the systemic inflammatory response syndrome (SIRS)[J]. Int J Mol Sci, 2023, 24(17). DOI:10.3390/ijms241713469 |
| [40] | Wei X, Tu Y, Bu S, et al. Unraveling the intricate web: complement activation shapes the pathogenesis of sepsis-Induced coagulopathy[J]. J Innate Immun, 2024, 16(1): 337-353. DOI:10.1159/000539502 |
| [41] | Ito T, Kakuuchi M, Maruyama I. Endotheliopathy in septic conditions: mechanistic insight into intravascular coagulation[J]. Crit Care, 2021, 25(1): 95. DOI:10.1186/s13054-021-03524-6 |
| [42] | Buchheim JI, Enzinger MC, Chouker A, et al. The stressed vascular barrier and coagulation - The impact of key glycocalyx components on in vitro clot formation[J]. Thromb Res, 2020, 186: 93-102. DOI:10.1016/j.thromres.2019.12.015 |
| [43] | Colicchia M, Perrella G, Gant P, et al. Novel mechanisms of thrombo-inflammation during infection: spotlight on neutrophil extracellular trap-mediated platelet activation[J]. Res Pract Thromb Haemost, 2023, 7(2): 100116. DOI:10.1016/j.rpth.2023.100116 |
| [44] | Lupu F, Kinasewitz G, Dormer K. The role of endothelial shear stress on haemodynamics, inflammation, coagulation and glycocalyx during sepsis[J]. J Cell Mol Med, 2020, 24(21): 12258-12271. DOI:10.1111/jcmm.15895 |
| [45] | Dolmatova EV, Wang K, Mandavilli R, et al. The effects of sepsis on endothelium and clinical implications[J]. Cardiovasc Res, 2021, 117(1): 60-73. DOI:10.1093/cvr/cvaa070 |
| [46] | Oshima K, King SI, Mcmurtry SA, et al. Endothelial heparan sulfate proteoglycans in sepsis: the role of the glycocalyx[J]. Semin Thromb Hemost, 2021, 47(3): 274-282. DOI:10.1055/s-0041-1725064 |
| [47] | Grassle S, Huck V, Pappelbaum KI, et al. von Willebrand factor directly interacts with DNA from neutrophil extracellular traps[J]. Arterioscler Thromb Vasc Biol, 2014, 34(7): 1382-1389. DOI:10.1161/ATVBAHA.113.303016 |
| [48] | Rahmati N, Keshavarz Motamed P, Maftoon N. Numerical study of ultra-large von Willebrand factor multimers in coagulopathy[J]. Biomech Model Mechanobiol, 2024, 23(3): 737-756. DOI:10.1007/s10237-023-01803-5 |
| [49] | Srdic T, Durasevic S, Lakic I, et al. From molecular mechanisms to clinical therapy: understanding sepsis-induced multiple organ dysfunction[J]. Int J Mol Sci, 2024, 25(14). DOI:10.3390/ijms25147770 |
| [50] | Huet O, Dupic L, Harrois A, et al. Oxidative stress and endothelial dysfunction during sepsis[J]. Front Biosci (Landmark Ed), 2011, 16(5): 1986-1995. DOI:10.2741/3835 |
| [51] | Joffre J, Hellman J. Oxidative Stress and endothelial dysfunction in sepsis and acute inflammation[J]. Antioxid Redox Signal, 2021, 35(15): 1291-1307. DOI:10.1089/ars.2021.0027 |
| [52] | Lopes-Pires ME, Frade-Guanaes JO, Quinlan GJ. Clotting dysfunction in sepsis: a role for ROS and potential for therapeutic intervention[J]. Antioxidants (Basel), 2021, 11(1). DOI:10.3390/antiox11010088 |
| [53] | Iba T, Maier CL, Tanigawa T, et al. Risk stratification utilizing sequential organ failure assessment (SOFA) score, antithrombin activity, and demographic data in sepsis-associated disseminated intravascular coagulation (DIC)[J]. Sci Rep, 2023, 13(1): 22502. DOI:10.1038/s41598-023-49855-y |
| [54] | Chappell D, Jacob M, Hofmann-Kiefer K, et al. Antithrombin reduces shedding of the endothelial glycocalyx following ischaemia/reperfusion[J]. Cardiovasc Res, 2009, 83(2): 388-396. DOI:10.1093/cvr/cvp097 |
| [55] | Sharma S, Tyagi T, Antoniak S. Platelet in thrombo-inflammation: Unraveling new therapeutic targets[J]. Front Immunol, 2022, 13: 1039843. DOI:10.3389/fimmu.2022.1039843 |
| [56] | Akahoshi T, Kaku N, Shono Y, et al. Impact of antithrombin activity levels following recombinant antithrombin gamma therapy in patients with sepsis-induced disseminated intravascular coagulation[J]. Clin Appl Thromb Hemost, 2022, 28: 10760296221135790. DOI:10.1177/10760296221135790 |
| [57] | Rodgers GM, Mahajerin A. Antithrombin therapy: current state and future outlook[J]. Clin Appl Thromb Hemost, 2023, 29: 10760296231205279. DOI:10.1177/10760296231205279 |
| [58] | Mohseni Afshar Z, Tavakoli Pirzaman A, Hosseinzadeh R, et al. Anticoagulant therapy in COVID-19: A narrative review[J]. Clin Transl Sci, 2023, 16(9): 1510-1525. DOI:10.1111/cts.13569 |
| [59] | Iba T, Tanigawa T, Wada H, et al. The antithrombin activity recovery after substitution therapy is associated with improved 28-day mortality in patients with sepsis-associated disseminated intravascular coagulation[J]. Thromb J, 2023, 21(1): 112. DOI:10.1186/s12959-023-00556-6 |
| [60] | Yarimizu K, Nakane M, Onodera Y, et al. Prognostic value of antithrombin activity levels in the early phase of intensive care: a 2-center retrospective cohort study[J]. Clin Appl Thromb Hemost, 2023, 29: 10760296231218711. DOI:10.1177/10760296231218711 |
| [61] | Thielen O, Mitra S, Debot M, et al. Mitigation of trauma-induced endotheliopathy by activated protein C: A potential therapeutic for postinjury thromboinflammation[J]. J Trauma Acute Care Surg, 2024, 96(1): 116-122. DOI:10.1097/TA.0000000000004142 |
| [62] | Oto J, Fernandez-Pardo A, Miralles M, et al. Activated protein C assays: A review[J]. Clin Chim Acta, 2020, 502: 227-232. DOI:10.1016/j.cca.2019.11.005 |
| [63] | Yong J, Abrams ST, Wang G, et al. Cell-free histones and the cell-based model of coagulation[J]. J Thromb Haemost, 2023, 21(7): 1724-1736. DOI:10.1016/j.jtha.2023.04.018 |
| [64] | Catenacci V, Sheikh F, Patel K, et al. The prognostic utility of protein C as a biomarker for adult sepsis: a systematic review and meta-analysis[J]. Crit Care, 2022, 26(1): 21. DOI:10.1186/s13054-022-03889-2 |
| [65] | Tanratana P, Sachetto ATA, Mast AE, et al. An anti-tissue factor pathway inhibitor antibody increases tissue factor activity in extracellular vesicles isolated from human plasma[J]. Res Pract Thromb Haemost, 2024, 8(1): 102275. DOI:10.1016/j.rpth.2023.102275 |
| [66] | Lupu C, Herlea O, Tang H, et al. Plasmin-dependent proteolysis of tissue factor pathway inhibitor in a mouse model of endotoxemia[J]. J Thromb Haemost, 2013, 11(1): 142-148. DOI:10.1111/jth.12044 |
| [67] | Ravindranath TM, Goto M, Iqbal O, et al. Plasma thrombin activatable fibrinolysis inhibitor and tissue factor pathway inhibitor changes following sepsis[J]. Clin Appl Thromb Hemost, 2007, 13(4): 362-368. DOI:10.1177/1076029607305580 |
| [68] | Iba T, Kidokoro A, Fukunaga M, et al. Association between the severity of sepsis and the changes in hemostatic molecular markers and vascular endothelial damage markers[J]. Shock, 2005, 23(1): 25-29. DOI:10.1097/01.shk.0000144422.32647.b6 |
| [69] | Madoiwa S, Nunomiya S, Ono T, et al. Plasminogen activator inhibitor 1 promotes a poor prognosis in sepsis-induced disseminated intravascular coagulation[J]. Int J Hematol, 2006, 84(5): 398-405. DOI:10.1532/IJH97.05190 |
| [70] | Takahashi H, Tatewaki W, Wada K, et al. Thrombin vs. plasmin generation in disseminated intravascular coagulation associated with various underlying disorders[J]. Am J Hematol, 1990, 33(2): 90-95. DOI:10.1002/ajh.2830330204 |
| [71] | Mutch NJ, Koikkalainen JS, Fraser SR, et al. Model thrombi formed under flow reveal the role of factor XⅢ-mediated cross-linking in resistance to fibrinolysis[J]. J Thromb Haemost, 2010, 8(9): 2017-2024. DOI:10.1111/j.1538-7836.2010.03963.x |
| [72] | Levi M, Van Der Poll T. Coagulation and sepsis[J]. Thromb Res, 2017, 149: 38-44. DOI:10.1016/j.thromres.2016.11.007 |
| [73] | Vago JP, Zaidan I, Perucci LO, et al. Plasmin and plasminogen prevent sepsis severity by reducing neutrophil extracellular traps and systemic inflammation[J]. JCI Insight, 2023, 8(8). DOI:10.1172/jci.insight.166044 |
| [74] | Hoshino K, Nakashio M, Maruyama J, et al. Validating plasminogen activator inhibitor-1 as a poor prognostic factor in sepsis[J]. Acute Med Surg, 2020, 7(1): e581. DOI:10.1002/ams2.581 |
| [75] | Morrow GB, Mutch NJ. Past, present, and future perspectives of plasminogen activator inhibitor 1 (PAI-1)[J]. Semin Thromb Hemost, 2023, 49(3): 305-313. DOI:10.1055/s-0042-1758791 |
| [76] | Komorowicz E, Farkas VJ, Szabo L, et al. DNA and histones impair the mechanical stability and lytic susceptibility of fibrin formed by staphylocoagulase[J]. Front Immunol, 2023, 14: 1233128. DOI:10.3389/fimmu.2023.1233128 |
| [77] | Zhou Y, Tao W, Shen F, et al. The emerging role of neutrophil extracellular traps in arterial, venous and cancer-associated thrombosis[J]. Front Cardiovasc Med, 2021, 8: 786387. DOI:10.3389/fcvm.2021.786387 |
| [78] | Watanabe-Kusunoki K, Nakazawa D, Ishizu A, et al. Thrombomodulin as a physiological modulator of intravascular injury[J]. Front Immunol, 2020, 11: 575890. DOI:10.3389/fimmu.2020.575890 |
| [79] | Aklilu A, Lai MS, Jiang Z, et al. Immunothrombosis in sepsis: cellular crosstalk, molecular triggers, and therapeutic opportunities-a review[J]. Int J Mol Sci, 2025, 26(13). DOI:10.3390/ijms26136114 |
| [80] | Engelmann B, Massberg S. Thrombosis as an intravascular effector of innate immunity[J]. Nat Rev Immunol, 2013, 13(1): 34-45. DOI:10.1038/nri3345 |
| [81] | Iba T, Helms J, Levi M, et al. Thromboinflammation in acute injury: infections, heatstroke, and trauma[J]. J Thromb Haemost, 2024, 22(1): 7-22. DOI:10.1016/j.jtha.2023.07.020 |
| [82] | Perez-Torres I, Aisa-Alvarez A, Casarez-Alvarado S, et al. Impact of treatment with antioxidants as an adjuvant to standard therapy in patients with septic shock: analysis of the correlation between cytokine storm and oxidative stress and therapeutic effects[J]. Int J Mol Sci, 2023, 24(23). DOI:10.3390/ijms242316610 |
| [83] | Li X, Li L, Shi Y, et al. Different signaling pathways involved in the anti-inflammatory effects of unfractionated heparin on lipopolysaccharide-stimulated human endothelial cells[J]. J Inflamm (Lond), 2020, 17: 5. DOI:10.1186/s12950-020-0238-7 |
| [84] | Iba T, Helms J, Connors JM, et al. The pathophysiology, diagnosis, and management of sepsis-associated disseminated intravascular coagulation[J]. J Intensive Care, 2023, 11(1): 24. DOI:10.1186/s40560-023-00672-5 |
| [85] | Bode C, Weis S, Sauer A, et al. Targeting the host response in sepsis: current approaches and future evidence[J]. Crit Care, 2023, 27(1): 478. DOI:10.1186/s13054-023-04762-6 |
| [86] | Esmon CT. Targeting factor Xa and thrombin: impact on coagulation and beyond[J]. Thromb Haemost, 2014, 111(4): 625-633. DOI:10.1160/TH13-09-0730 |
| [87] | Ma L, Dorling A. The roles of thrombin and protease-activated receptors in inflammation[J]. Semin Immunopathol, 2012, 34(1): 63-72. DOI:10.1007/s00281-011-0281-9 |
| [88] | Stavenuiter F, Mosnier LO. Noncanonical PAR3 activation by factor Xa identifies a novel pathway for Tie2 activation and stabilization of vascular integrity[J]. Blood, 2014, 124(23): 3480-3489. DOI:10.1182/blood-2014-06-582775 |
| [89] | Strande JL, Phillips SA. Thrombin increases inflammatory cytokine and angiogenic growth factor secretion in human adipose cells in vitro[J]. J Inflamm (Lond), 2009, 6: 4. DOI:10.1186/1476-9255-6-4 |
| [90] | Minami T, Sugiyama A, Wu SQ, et al. Thrombin and phenotypic modulation of the endothelium[J]. Arterioscler Thromb Vasc Biol, 2004, 24(1): 41-53. DOI:10.1161/01.ATV.0000099880.09014.7D |
| [91] | Mosad E, Elsayh KI, Eltayeb AA. Tissue factor pathway inhibitor and P-selectin as markers of sepsis-induced non-overt disseminated intravascular coagulopathy[J]. Clin Appl Thromb Hemost, 2011, 17(1): 80-87. DOI:10.1177/1076029609344981 |
| [92] | Yang CC, Hsiao LD, Yang CM, et al. Thrombin enhanced matrix metalloproteinase-9 expression and migration of SK-N-SH cells via PAR-1, c-Src, PYK2, EGFR, Erk1/2 and AP-1[J]. Mol Neurobiol, 2017, 54(5): 3476-3491. DOI:10.1007/s12035-016-9916-0 |
| [93] | Wojta J. Macrophages and Thrombin-Another Link between Inflammation and Coagulation[J]. Thromb Haemost, 2020, 120(4): 537. DOI:10.1055/s-0040-1708551 |
| [94] | Rahman A, Fazal F. Hug tightly and say goodbye: role of endothelial ICAM-1 in leukocyte transmigration[J]. Antioxid Redox Signal, 2009, 11(4): 823-839. DOI:10.1089/ars.2008.2204 |
| [95] | Blackburn JS, Brinckerhoff CE. Matrix metalloproteinase-1 and thrombin differentially activate gene expression in endothelial cells via PAR-1 and promote angiogenesis[J]. Am J Pathol, 2008, 173(6): 1736-1746. DOI:10.2353/ajpath.2008.080512 |
| [96] | Laterre PF, Levy H, Clermont G, et al. Hospital mortality and resource use in subgroups of the Recombinant Human Activated Protein C Worldwide Evaluation in Severe Sepsis (PROWESS) trial[J]. Crit Care Med, 2004, 32(11): 2207-2218. DOI:10.1097/01.ccm.0000145231.71605.d8 |
| [97] | Ranieri Vm, Thompson Bt, Prowess-Shock Study Group, et al. Drotrecogin alfa (activated) in adults with septic shock[J]. N Engl J Med, 2012, 366(22): 2055-2064. DOI:10.1056/NEJMoa1202290 |
| [98] | Holder Al, Dt H. A dream deferred: the rise and fall of recombinant activated protein C[J]. Crit Care, 2013, 17(2): 309. DOI:10.1186/cc12533 |
| [99] | Gardlund B, Dmitrieva NO, Pieper CF, et al. Six subphenotypes in septic shock: Latent class analysis of the PROWESS Shock study[J]. J Crit Care, 2018, 47: 70-79. DOI:10.1016/j.jcrc.2018.06.012 |
| [100] | Ely Ew, Laterre Pf, Angus Dc, et al. Drotrecogin alfa (activated) administration across clinically important subgroups of patients with severe sepsis[J]. Crit Care Med, 2003, 31(1): 12-19. DOI:10.1097/01.CCM.0000046070.25223.79-Crit |
| [101] | Wiedermann CJ, Hoffmann JN, Juers M, et al. High-dose antithrombin Ⅲ in the treatment of severe sepsis in patients with a high risk of death: efficacy and safety[J]. Crit Care Med, 2006, 34(2): 285-292. DOI:10.1097/01.ccm.0000194731.08896.99 |
| [102] | Yamakawa K, Aihara M, Ogura H, et al. Recombinant human soluble thrombomodulin in severe sepsis: a systematic review and meta-analysis[J]. J Thromb Haemost, 2015, 13(4): 508-519. DOI:10.1111/jth.12841 |
| [103] | Yoshimura J, Yamakawa K, Ogura H, et al. Benefit profile of recombinant human soluble thrombomodulin in sepsis-induced disseminated intravascular coagulation: a multicenter propensity score analysis[J]. Crit Care, 2015, 19(1): 78. DOI:10.1186/s13054-015-0810-3 |
| [104] | Iba T, Nisio MD, Levy JH, et al. New criteria for sepsis-induced coagulopathy (SIC) following the revised sepsis definition: a retrospective analysis of a nationwide survey[J]. BMJ Open, 2017, 7(9): e017046. DOI:10.1136/bmjopen-2017-017046 |
| [105] | Iba T, Levy JH, Thachil J, et al. The progression from coagulopathy to disseminated intravascular coagulation in representative underlying diseases[J]. Thromb Res, 2019, 179: 11-14. DOI:10.1016/j.thromres.2019.04.030 |
| [106] | Iba T, Levy JH, Scientific and Standardization Committee on Dic of the International Society on Thrombosis and Haemostasis, et al. Proposal of a two-step process for the diagnosis of sepsis-induced disseminated intravascular coagulation[J]. J Thromb Haemost, 2019, 17(8): 1265-1268. DOI:10.1111/jth.14482 |
| [107] | Iba T, Arakawa M, Di Nisio M, et al. Newly proposed sepsis-induced coagulopathy precedes international society on thrombosis and haemostasis overt-disseminated intravascular coagulation and predicts high mortality[J]. J Intensive Care Med, 2020, 35(7): 643-649. DOI:10.1177/0885066618773679 |
| [108] | Curtiaud A, Iba T, Angles-Cano E, et al. Biomarkers of sepsis-induced coagulopathy: diagnostic insights and potential therapeutic implications[J]. Ann Intensive Care, 2025, 15(1): 12. DOI:10.1186/s13613-025-01434-2 |
| [109] | Kato H, Hagihara M, Asai N, et al. Efficacy and safety of recombinant human soluble thrombomodulin in patients with sepsis-induced disseminated intravascular coagulation - A meta-analysis[J]. Thromb Res, 2023, 226: 165-172. DOI:10.1016/j.thromres.2023.05.009 |
| [110] | Yamakawa K, Fujimi S, Mohri T, et al. Treatment effects of recombinant human soluble thrombomodulin in patients with severe sepsis: a historical control study[J]. Crit Care, 2011, 15(3): R123. DOI:10.1186/cc10228 |
| [111] | Ushio N, Yamakawa K, Mochizuki K, et al. Efficacy of unfractionated heparin in patients with moderate sepsis-induced coagulopathy: An observational study[J]. Thromb Res, 2024, 241: 109095. DOI:10.1016/j.thromres.2024.109095 |
| [112] | Yamakawa K, Umemura Y, Hayakawa M, et al. Benefit profile of anticoagulant therapy in sepsis: a nationwide multicentre registry in Japan[J]. Crit Care, 2016, 20(1): 229. DOI:10.1186/s13054-016-1415-1 |
| [113] | Yamakawa K, Yoshimura J, Ito T, et al. External validation of the two newly proposed criteria for assessing coagulopathy in sepsis[J]. Thromb Haemost, 2019, 119(2): 203-212. DOI:10.1055/s-0038-1676610 |
| [114] | Totoki T, Koami H, Makino Y, et al. Heparin therapy in sepsis and sepsis-associated disseminated intravascular coagulation: a systematic review and meta-analysis[J]. Thromb J, 2024, 22(1): 84. DOI:10.1186/s12959-024-00653-0 |
| [115] | Rhodes A, Evans LE, Alhazzani W, et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016[J]. Crit Care Med, 2017, 45(3): 486-552. DOI:10.1097/CCM.0000000000002255 |
| [116] | Abraham E, Reinhart K, Opal S, et al. Efficacy and safety of tifacogin (recombinant tissue factor pathway inhibitor) in severe sepsis: a randomized controlled trial[J]. JAMA, 2003, 290(2): 238-247. DOI:10.1001/jama.290.2.238 |
| [117] | Saito H, Maruyama I, Shimazaki S, et al. Efficacy and safety of recombinant human soluble thrombomodulin (ART-123) in disseminated intravascular coagulation: results of a phase Ⅲ, randomized, double-blind clinical trial[J]. J Thromb Haemost, 2007, 5(1): 31-41. DOI:10.1111/j.1538-7836.2006.02267.x |
| [118] | Tejada S, Clemente A, Socias A, et al. Circulating NETs enable early identification of thrombotic risk in sepsis at emergency care onset[J]. Front Immunol, 2025, 16: 1664108. DOI:10.3389/fimmu.2025.1664108 |
| [119] | Lindahl U, Li JP. Heparin - An old drug with multiple potential targets in Covid-19 therapy[J]. J Thromb Haemost, 2020, 18(9): 2422-2424. DOI:10.1111/jth.14898 |
| [120] | Hogwood J, Gray E, Mulloy B. Heparin, heparan sulphate and sepsis: potential new options for treatment[J]. Pharmaceuticals (Basel), 2023, 16(2). DOI:10.3390/ph16020271 |
| [121] | Fu S, Yu S, Zhao Y, et al. Unfractionated heparin attenuated histone-induced pulmonary syndecan-1 degradation in mice: a preliminary study on the roles of heparinase pathway[J]. Inflammation, 2022, 45(2): 712-724. DOI:10.1007/s10753-021-01578-w |
| [122] | Galli E, Maggio E, Pomero F. Venous Thromboembolism in sepsis: from bench to bedside[J]. Biomedicines, 2022, 10(7). DOI:10.3390/biomedicines10071651 |
| [123] | Wildhagen KC, Garcia De Frutos P, Reutelingsperger CP, et al. Nonanticoagulant heparin prevents histone-mediated cytotoxicity in vitro and improves survival in sepsis[J]. Blood, 2014, 123(7): 1098-1101. DOI:10.1182/blood-2013-07-514984 |
| [124] | Wang P, Chi L, Zhang Z, et al. Heparin: An old drug for new clinical applications[J]. Carbohydr Polym, 2022, 295: 119818. DOI:10.1016/j.carbpol.2022.119818 |
| [125] | Ebeyer-Masotta M, Eichhorn T, Weiss R, et al. Heparin-functionalized adsorbents eliminate central effectors of immunothrombosis, including platelet factor 4, high-mobility group box 1 protein and histones[J]. Int J Mol Sci, 2022, 23(3). DOI:10.3390/ijms23031823 |
| [126] | Huckriede JB, Beurskens DMH, Wildhagen K, et al. Design and characterization of novel activated protein C variants for the proteolysis of cytotoxic extracellular histone H3[J]. J Thromb Haemost, 2023, 21(12): 3557-3567. DOI:10.1016/j.jtha.2023.08.023 |
| [127] | Huang X, Han S, Liu X, et al. Both UFH and NAH alleviate shedding of endothelial glycocalyx and coagulopathy in LPS-induced sepsis[J]. Exp Ther Med, 2020, 19(2): 913-922. DOI:10.3892/etm.2019.8285 |
| [128] | Sharma N, Haggstrom L, Sohrabipour S, et al. Investigations of the effectiveness of heparin variants as inhibitors of histones[J]. J Thromb Haemost, 2022, 20(6): 1485-1495. DOI:10.1111/jth.15706 |
| [129] | Iba T, Saitoh D. Efficacy of antithrombin in preclinical and clinical applications for sepsis-associated disseminated intravascular coagulation[J]. J Intensive Care, 2014, 2(1): 66. DOI:10.1186/s40560-014-0051-6 |
| [130] | Kirschenbaum LA, Lopez WC, Ohrum P, et al. Effect of recombinant activated protein C and low-dose heparin on neutrophil-endothelial cell interactions in septic shock[J]. Crit Care Med, 2006, 34(8): 2207-2212. DOI:10.1097/01.CCM.0000229880.41513.86 |
| [131] | Altaweel L, Sweeney D, Cui X, et al. Growing insights into the potential benefits and risks of activated protein C administration in sepsis: a review of preclinical and clinical studies[J]. Biologics, 2009, 3: 391-406. DOI:10.2147/btt.2009.3547 |
| [132] | Annane D, Timsit JF, Megarbane B, et al. Recombinant human activated protein C for adults with septic shock: a randomized controlled trial[J]. Am J Respir Crit Care Med, 2013, 187(10): 1091-1097. DOI:10.1164/rccm.201211-2020OC |
| [133] | Annane D, Buisson CB, Cariou A, et al. Design and conduct of the activated protein C and corticosteroids for human septic shock (APROCCHSS) trial[J]. Ann Intensive Care, 2016, 6(1): 43. DOI:10.1186/s13613-016-0147-3 |
| [134] | Mosnier LO, Gale AJ, Yegneswaran S, et al. Activated protein C variants with normal cytoprotective but reduced anticoagulant activity[J]. Blood, 2004, 104(6): 1740-1744. DOI:10.1182/blood-2004-01-0110 |
| [135] | Flaumenhaft R, De Ceunynck K. Targeting PAR1: now what?[J]. Trends Pharmacol Sci, 2017, 38(8): 701-716. DOI:10.1016/j.tips.2017.05.001 |
| [136] | Abraham E, Laterre Pf, Garg R, et al. Drotrecogin alfa (activated) for adults with severe sepsis and a low risk of death[J]. N Engl J Med, 2005, 353(13): 1332-1341. DOI:10.1056/NEJMoa050935 |
| [137] | Papageorgiou C, Jourdi G, Adjambri E, et al. Disseminated intravascular coagulation: an update on pathogenesis, diagnosis, and therapeutic strategies[J]. Clin Appl Thromb Hemost, 2018, 24(9_suppl): 8S-28S. DOI:10.1177/1076029618806424 |
| [138] | Levy M, Levi M, Williams MD, et al. Comprehensive safety analysis of concomitant drotrecogin alfa (activated) and prophylactic heparin use in patients with severe sepsis[J]. Intensive Care Med, 2009, 35(7): 1196-1203. DOI:10.1007/s00134-009-1483-7 |
| [139] | Cox D. Sepsis - it is all about the platelets[J]. Front Immunol, 2023, 14: 1210219. DOI:10.3389/fimmu.2023.1210219 |
| [140] | Giustozzi M, Ehrlinder H, Bongiovanni D, et al. Coagulopathy and sepsis: Pathophysiology, clinical manifestations and treatment[J]. Blood Rev, 2021, 50: 100864. DOI:10.1016/j.blre.2021.100864 |
| [141] | Li X, Zhang G, Cao X. The function and regulation of platelet P2Y12 receptor[J]. Cardiovasc Drugs Ther, 2023, 37(1): 199-216. DOI:10.1007/s10557-021-07229-4 |
| [142] | Greco E, Lupia E, Bosco O, et al. Platelets and multi-organ failure in sepsis[J]. Int J Mol Sci, 2017, 18(10). DOI:10.3390/ijms18102200 |
| [143] | Muhlestein JB. Effect of antiplatelet therapy on inflammatory markers in atherothrombotic patients[J]. Thromb Haemost, 2010, 103(1): 71-82. DOI:10.1160/TH09-03-0177 |
| [144] | Akinosoglou K, Alexopoulos D. Use of antiplatelet agents in sepsis: a glimpse into the future[J]. Thromb Res, 2014, 133(2): 131-138. DOI:10.1016/j.thromres.2013.07.002 |
| [145] | Ouyang Y, Wang Y, Liu B, et al. Effects of antiplatelet therapy on the mortality rate of patients with sepsis: A meta-analysis[J]. J Crit Care, 2019, 50: 162-168. DOI:10.1016/j.jcrc.2018.12.004 |
| [146] | Wang Y, Ouyang Y, Liu B, et al. Platelet activation and antiplatelet therapy in sepsis: A narrative review[J]. Thromb Res, 2018, 166: 28-36. DOI:10.1016/j.thromres.2018.04.007 |
| [147] | Al-Husinat L, Abu Hmaid A, Abbas H, et al. Role of aspirin, beta-blocker, statins, and heparin therapy in septic patients under mechanical ventilation: a narrative review[J]. Front Med (Lausanne), 2023, 10: 1143090. DOI:10.3389/fmed.2023.1143090 |
| [148] | Kobayashi M, Kudo D, Ohbe H, et al. Antiplatelet pretreatment and mortality in patients with severe sepsis: A secondary analysis from a multicenter, prospective survey of severe sepsis in Japan[J]. J Crit Care, 2022, 69: 154015. DOI:10.1016/j.jcrc.2022.154015 |
| [149] | Hsu WT, Porta L, Chang IJ, et al. Association between aspirin use and sepsis outcomes: a national cohort study[J]. Anesth Analg, 2022, 135(1): 110-117. DOI:10.1213/ANE.0000000000005943 |
 2026, Vol. 35
2026, Vol. 35